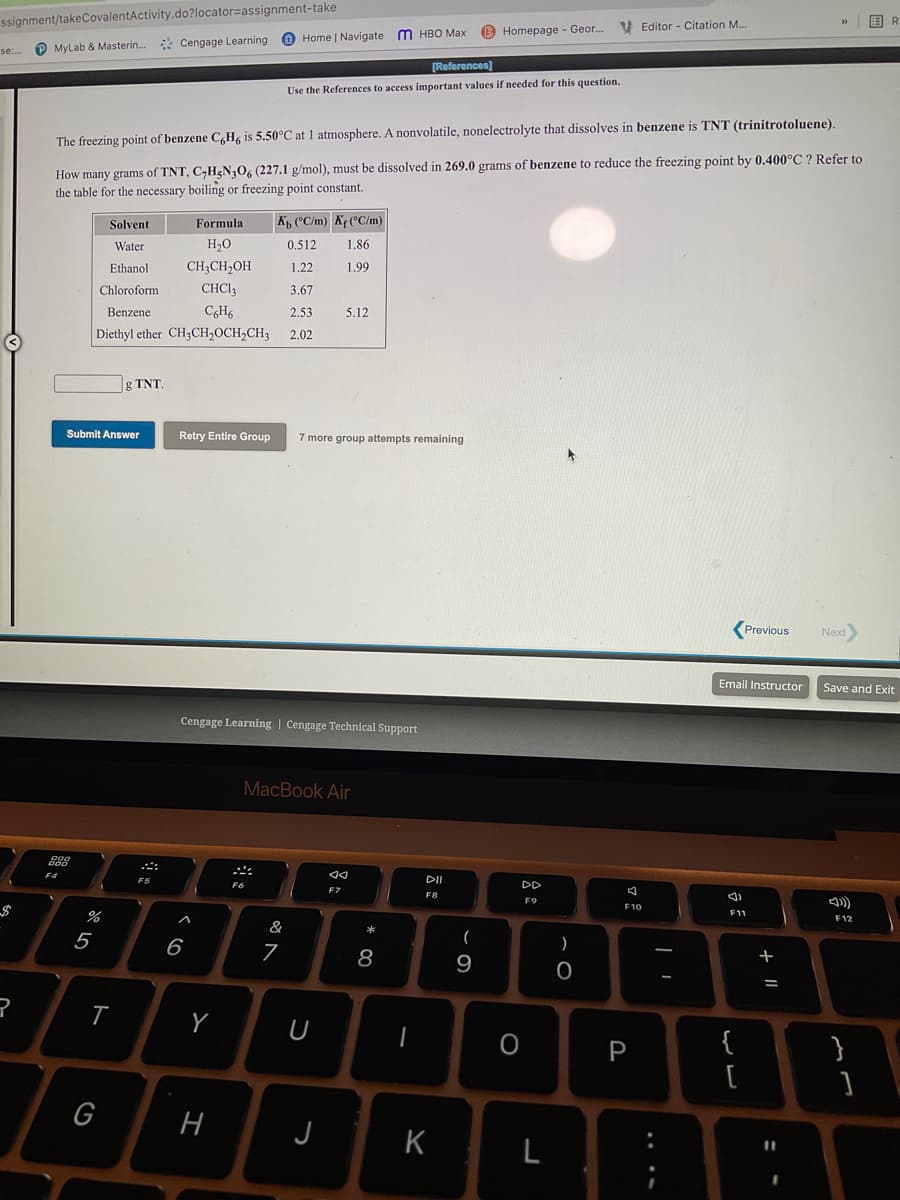
The freezing point of benzene C,H, is 5.50°C at 1 atmosphere. A nonvolatile, nonelectrolyte that dissolves in benzene is TNT (trinitrotoluene). How many grams of TNT, C,H&N3O6 (227.1 g/mol), must be dissolved in 269.0 grams of benzene to reduce the freezing point by 0.400°C ? Refer to the table for the necessary boiling or freezing point constant. Solvent Formula K CC/m) KrCC/m) Water H,0 0.512 1.86 Ethanol CH;CH,OH 1.22 1.99 Chloroform CHCI3 3.67 Benzene CH6 2.53 5.12 Diethyl ether CH3CH2OCH2CH3 2.02
The freezing point of benzene C,H, is 5.50°C at 1 atmosphere. A nonvolatile, nonelectrolyte that dissolves in benzene is TNT (trinitrotoluene). How many grams of TNT, C,H&N3O6 (227.1 g/mol), must be dissolved in 269.0 grams of benzene to reduce the freezing point by 0.400°C ? Refer to the table for the necessary boiling or freezing point constant. Solvent Formula K CC/m) KrCC/m) Water H,0 0.512 1.86 Ethanol CH;CH,OH 1.22 1.99 Chloroform CHCI3 3.67 Benzene CH6 2.53 5.12 Diethyl ether CH3CH2OCH2CH3 2.02
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter11: Properties Of Solutions
Section: Chapter Questions
Problem 28Q
Related questions
Question
I don’t know how to do this question
Transcribed Image Text:ssignment/takeCovalentActivity.do?locator=assignment-take
国R
B Homepage - Geor. Editor - Citation M...
Home | Navigate m HBO Max
MyLab & Masterin. * Cengage Learning
se:.
[References)
Use the References to access important values if needed for this question.
The freezing point of benzene C,H, is 5.50°C at 1 atmosphere. A nonvolatile, nonelectrolyte that dissolves in benzene is TNT (trinitrotoluene).
How many grams of TNT, C,H<N30, (227.1 g/mol), must be dissolved in 269.0 grams of benzene to reduce the freezing point by 0.400°C ? Refer to
the table for the necessary boiling or freezing point constant.
Solvent
Formula
K, (°C/m) Kr (°C/m)
Water
H2O
0.512
1.86
Ethanol
CH;CH,OH
1.22
1.99
Chloroform
CHCI3
3.67
Benzene
CH6
2.53
5.12
Diethyl ether CH3CH,0CH,CH3
2.02
g TNT.
Submit Answer
Retry Entire Group
7 more group attempts remaining
Previous
Next
Email Instructor
Save and Exit
Cengage Learning | Cengage Technical Support
MacBook Air
F4
DII
DD
F7
F8
F9
F10
F11
F12
&
5
6
7
)
8
9
+
T
Y
{
}
[
G
H
J
K
L
+ ||
....
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax