The Group 2 metals (Ba, Ca, Sr) can be produced in the elemental state by the reaction of their oxides with aluminum metal at high temperatures, also producing solid aluminum oxide as a by-product Choose the unbalanced chemical equation for the reaction of barium oxide with aluminum. O Ba0(s) + Ba() Al(s) + Al, O3(a) O BaO(s) + Al(s) + Ba(s) + Ala0;(s) O Ba(a) + Al(s) Ba0(a) + Al O,(a) O Ba(a) + Al,O,(a) Ba0(s) + AI(s) b Choose the unbalanced chemical equation for the reaction of calcium oxide with aluminum. O CaO(s) + Ca(s) Al(a) + AlO,(s) O CaO(a) + Al() Ca(s) + AlO,(s) O Ca(s) + Al(s) CaO(s) + Al;Os() O Ca(a) + Al2O(s)CaO(s)+Al(s) Choose the unbalanced chemical equation for the reaction of strontium oxide with aluminum. O Sro(s) + Sr(s)+ Al(s) + AlO (s) O SrO(a) + Al(s) Sr(s) + Al Og(s) O Sr(a) + Al(a) SrO(a) + AlO3(a) O Sr(s) + AlO,(s) SrO(s) + Al(s)
The Group 2 metals (Ba, Ca, Sr) can be produced in the elemental state by the reaction of their oxides with aluminum metal at high temperatures, also producing solid aluminum oxide as a by-product Choose the unbalanced chemical equation for the reaction of barium oxide with aluminum. O Ba0(s) + Ba() Al(s) + Al, O3(a) O BaO(s) + Al(s) + Ba(s) + Ala0;(s) O Ba(a) + Al(s) Ba0(a) + Al O,(a) O Ba(a) + Al,O,(a) Ba0(s) + AI(s) b Choose the unbalanced chemical equation for the reaction of calcium oxide with aluminum. O CaO(s) + Ca(s) Al(a) + AlO,(s) O CaO(a) + Al() Ca(s) + AlO,(s) O Ca(s) + Al(s) CaO(s) + Al;Os() O Ca(a) + Al2O(s)CaO(s)+Al(s) Choose the unbalanced chemical equation for the reaction of strontium oxide with aluminum. O Sro(s) + Sr(s)+ Al(s) + AlO (s) O SrO(a) + Al(s) Sr(s) + Al Og(s) O Sr(a) + Al(a) SrO(a) + AlO3(a) O Sr(s) + AlO,(s) SrO(s) + Al(s)
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter4: Stoichiometry
Section: Chapter Questions
Problem 4.68PAE: 4.68 The pictures below show a molecular-scale view of a chemical reaction between the compounds AB2...
Related questions
Question
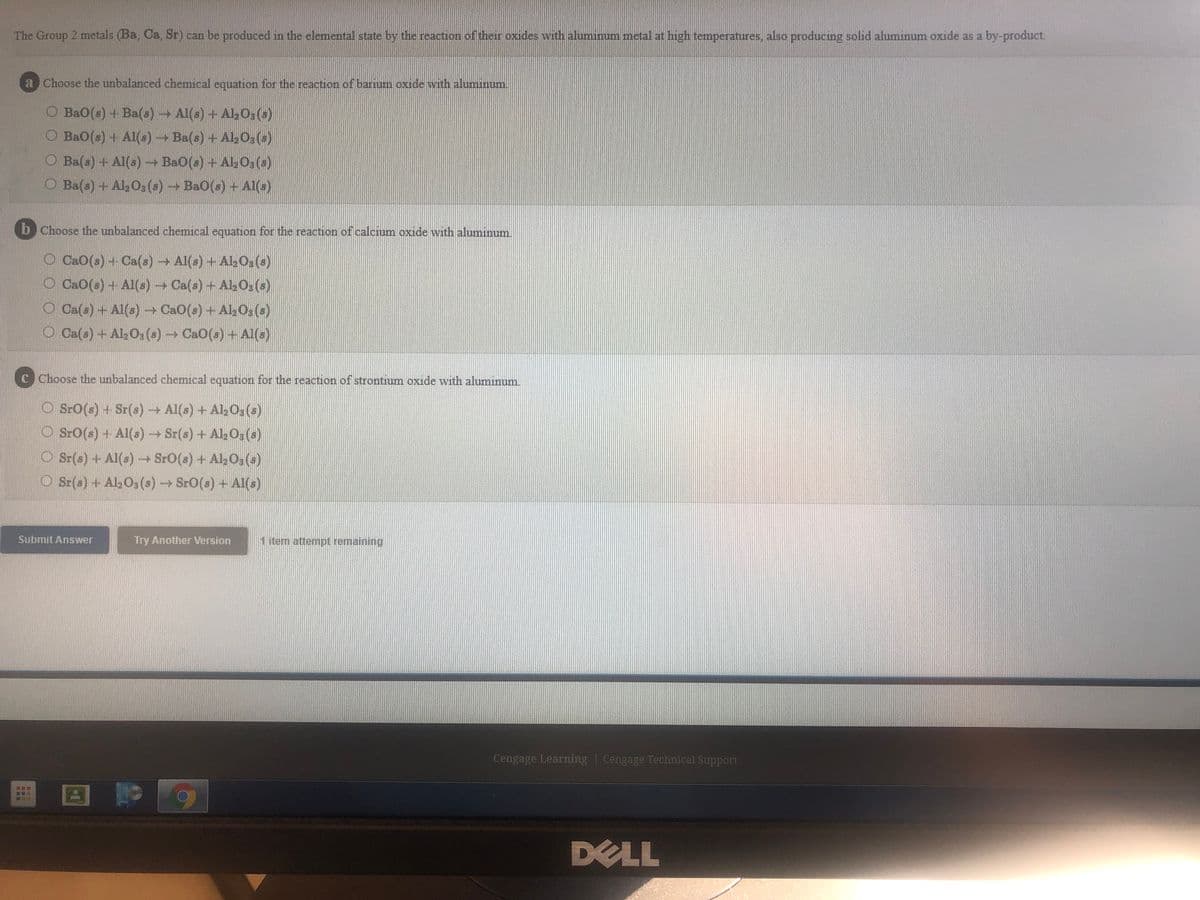
Transcribed Image Text:The Group 2 metals (Ba, Ca, Sr) can be produced in the elemental state by the reaction of their oxides with aluminum metal at high temperatures, also producing solid aluminum oxide as a by-product.
a Choose the unbalanced chemical equation for the reaction of barum oxide with aluminum.
O Ba0(s) + Ba(s) Al(s) + Al03 (s)
Ba0(s) + Al(s)- Ba(s) + Al, O3 (a)
O Ba(s) + Al(s) Ba0(s) + Al,0 (a)
O Ba(s) + Al,O3 (s) Ba0(s) + Al(s)
b Choose the unbalanced chemical equation for the reaction of calcium oxide with aluminum.
O CaO(s) + Ca(s)Al(s) + Al203 (s)
O Ca0(s) + Al(s) → Ca(s) +Ak0: (s)
O Ca(s) + Al(s) CaO(s) + Al, 0%(s)
O Ca(s) + Al2O; (s) CaO(s) + AI(s)
C Choose the unbalanced chemical equation for the reaction of strontium oxide with aluminum
O SrO(s) + Sr(s) Al(s) + Al, 0:(s)
O Sr0(s) + Al(s) Sr(s) + Al,O, (s)
O Sr(s) + Al(s) SrO(s) + Al, O, (3)
O Sr(s) + Al, O3 (s)SrO(s) + Al(s)
Submit Answer
Try Another Version
1 item attempt remaining
Cengage Learning | Cengage Technical Support
DELL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning