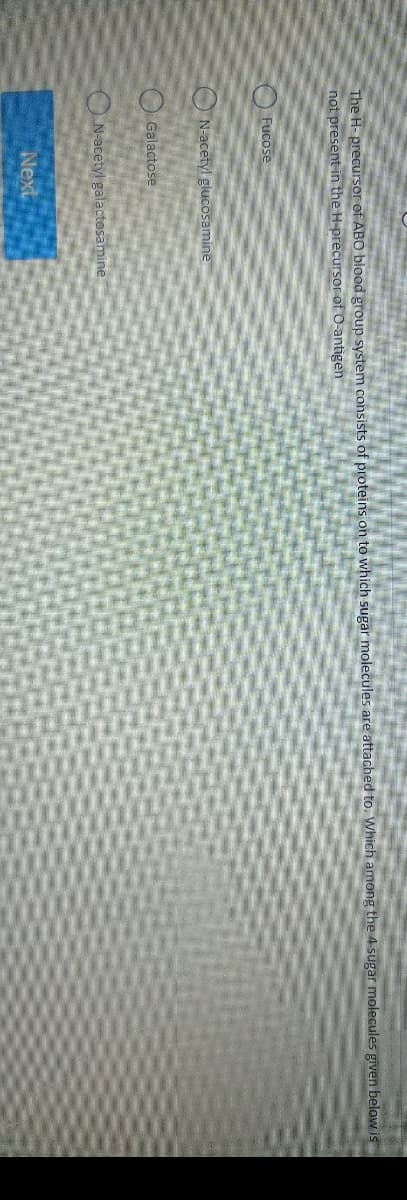
The H-precursor of ABO blood group system consists of proteins on to which sugar moles not present in the H-precursor of O-antigen Fucose N-acetyl glucosamine Galactose N-acetyl galactosamine O
Q: What is the total number of hydrogen bonds that exist between the DNA strand 5’-TTCAGAG-3’ and its…
A: Adenine and guanine are the purine bases which occur in the nucleic acids. And thymine, cytosine,…
Q: What is the primary reason the a-helix conformation in polypeptides such a stable form? A. The…
A: The secondary structure of protein determines the local spatial arrangement of the amino acids in…
Q: A student Hussein woke up early and then went quickly - without having breakfast - to Al-Maaqal…
A: Glucose is a very important source of energy & it is required by our body to carry out normal…
Q: A protein was recently discovered to be located in the nucleus. However, it is uncertain whether…
A: The most important and basic knowledge that we should remember here in-order to understand the…
Q: Draw the structure of 1-linolenyl-2-arachidyl-3-phosphatidylserine
A: Phosphatidyl serine is a glycerophospholipid composed of fatty acids, phosphate group, serine, and a…
Q: The reaction of glutamate and NH4* to yield glutamine is catalyzed by a) Uridylyltransferase b)…
A: Glutamine and glutamate are important amino acids for the metabolism of body. Though they are not…
Q: The image below shows the different interactions responsible for the spontaneous folding of a…
A: Protein protein interactions are of different kinds, they are responsible for holding the protein…
Q: 1) Dilution fraction 2) Water
A: Dilution is a method to dilute the specific sample to achieve the required low concentration of the…
Q: The "energy balance model" of body mass is based on the idea that biological systems neither create…
A: Energy intake, energy expenditure, and energy storage are the three basic components of energy…
Q: Why are nucleic acids considered a non-essential component of the diet? Bases, specifically purines…
A: Nucleic acids are biomolecules composed of nucleotide units. Each nucleotide unit is composed of a…
Q: How much ATP is produced from 1-stearoyl-2-arachidonyl-3-tridecanoyl glyceride? Show complete…
A: The given triglyceride molecule will be converted to stearic acid, arachidonic acid, tridecanoic…
Q: 1) under intracellular conditions, answer : If G3P-DH is inhibited by Iodoacetic acid, which…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: toxin) njected, it paralyze locally. Based on this information only, what neurotransmitter system…
A: Botox (Botulinum toxin)is considered as a neurotoxin protein ,this toxin can lead to a condition…
Q: the correct amount that can be resolved from the TLC?
A: There are apperenttly 8 Isomers of ketohexoses which are D-Fructose, D-tagatose, D-psicose, and…
Q: Glycolysis consists of three irreversible steps. Which of the following enzyme- catalyzed reaction…
A: Glycolysis is the first stage of cellular respiration during which glucose is converted to pyruvate…
Q: Show, by hand drawing a diagram, how a disaccharide and polysaccharide is formed
A: Polysaccharides are made up of a large number of monomers, whereas disaccharides are made up of two…
Q: When part of a nucleotide in a nucleic acid chain, which of the following may base pair with uridine…
A: Uridine is a major form of pyrimidine nucleotide, from which cytosine and uracil are derived. It is…
Q: Modify isoleucine to show the predominant forms at pH 1, 7, and 13. Isoleucine has pK, values of 2.4…
A: Proteins are polymer of amino acids , and each of the amino acid residue is linked to its…
Q: what is the purpose of this experiment Effect of Temperature on the Hydrolysis of Starc
A: Introduction: Enzymes are proteins that act as biological catalysts to fasten the rate of the…
Q: Peptide 1: Peptide 2: NH₂ H₂N pro-ile-glu-arg N OH OH OH OH
A:
Q: The following peptide is cut by serine protease enzyme Trypsin. How many fragments will be produced…
A: Proteases are enzymes which digest proteins by cleaving the peptide bonds. Trypsin is a protease…
Q: How the different types of fats in one’s diet can help correct or prevent heart disease
A: Fat is an essential part of diet that have different important role in the body. Mainly these are of…
Q: In the double-reciprocal plot, increasing the concentration of inhibitor A. raises the Vmax. B.…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: Please help me I really a comprehensive explanation please help. Talk about covid and why it’s a…
A: Covid occurs by a very deadly virus that has the ability to affect different people in distinct…
Q: Why is it important to drink water immediately after taking aspirin? How is the stomach wall…
A: Aspirin (acetylsalicylic acid or ASA) is a type of nonsteroidal anti-inflammatory medication…
Q: What glycolytic intermediate is fructose converted to in the muscle, such that it can be utilized in…
A: Fructose is an abundant dietary monosaccharide that is present naturally in fruits and vegetables…
Q: Which of the following describe active transport or is an example of a transport of a substance…
A: Since you have posted a question with multiple questions, we will solve the first question for you.…
Q: Signaling pathways often require receptor dimers to become active. What would be an advantage of the…
A: Most receptors dimerize during their association with their ligands. This dimerization provides them…
Q: Which of the following mutagens results in the deamination of nitrogenous bases? O nitrous acid base…
A: The amino bases adenine and cytosine lose one amino group when they are oxidised. As a result, in…
Q: What is the role of tyrosine in prostaglandin synthesis? Tyrosine provides the proton to the double…
A: Prostaglandins are mostly produced from Arachidonic acids( a C20 poly-unsaturated fatty acids) in…
Q: Adenylylation is a post-translational modification that can regulate the Adenylyation involves the…
A: Introduction: In living organisms, hundreds of different enzyme-catalyzed reactions occur…
Q: You are talking with a winemaker about yeast. "I know that yeast can survive under both aerobic and…
A: Yeast can survive in the presence of oxygen as well as in the absence of oxygen. Wine is a…
Q: Question 5 (2 points) Which of the following decreases the affinity of hemoglobin for oxygen? Choo-…
A: There is a continuous relationship between the oxygen affinity of hemoglobin and oxygen saturation…
Q: From the Lineweaver-Burke plot of an enzyme-catalyzed reaction containing 4 μM total enzyme, you…
A: Hi. Thank you for the question. As per the honor code, We'll answer the first question since the…
Q: Using good details, compare and contrast the pairs of different biochemical reactions. Create your…
A: Introduction: The term metabolism describes the interconversion of chemical compounds in the body…
Q: 1 Chemistry of Life: Biomol Which of the following is NOT a carbon based molecule. O nucleotide O…
A: Introduction: Biomolecules are molecules of compounds that are needed for life. They occur naturally…
Q: During hydrogenation, cis double bonds are converted to trans double bonds. In the lab, we compare…
A: Cis and trans Fatty acids have hydrogen atoms organised in different manner where cis fatty acids…
Q: Which of the following factors is responsible for the denaturation of proteins? * Oa) Heat Ob)…
A: Denaturation of any protein includes a number of weak bonds or weak linkages, that occur within a…
Q: Why is the AT base pair less stable than the GC base pair
A: DNA is a genetic material present in most living organisms and it is composed of nucleotides.…
Q: Humans do not undergo net synthesis of carbohydrate from acetyl- CoA, yet carbons of acetyl -CoA can…
A: Since you have posted multiple questions we will answer the first question for you. If you want any…
Q: What is the structural difference between the pentose sugars in DNA and RNA?
A: Pentose sugar is a five carbon molecule numbered as 1', 2', 3', 4' and 5'. When the functional…
Q: The concentrations of pyruvate, NADH, H+, lactate, and NAD are 2, 1.5, 1.5, 1.2, 1.2 mm,…
A: Given, NADH : 1.5 mM H+ : 1.5 mM lactate : 1.2 mM NAD+ : 1.2 mM pyruvate: 2 mM also it is given…
Q: The base that is circled in red could be adenine thymine guanine None of the provided answer choices…
A: The molecular basis of heredity is DNA. It can be thought of as a genetic information reserve bank.…
Q: Name the monomers for all the macromolecules in Test Tubes 1-5 in Jenny's experiment. Benedict's…
A: Living organisms are constituted of four types of biological macromolecules nucleic acids, proteins,…
Q: The following enzyme has an exact integer number of turns. What is the length and amount of amino…
A: Enzyme are usually composed of proteins while some enzymes are ribozymes (RNA enzymes). Proteins are…
Q: 1A. The pyruvate dehydrogenase complex is heavily regulated as the nexus between glycolysis and the…
A: "Since you have asked multiple subparts of a questions, we will solve the first two subparts of the…
Q: O a collaboration between particular microbiomes and liver metabolism
A: TMAO is stand for the trimethylamine N-oxide and its small colorless amine oxide which generate from…
Q: What is the actual meaning of Biocompatible and biodegrable for the scaffolds for biomedical…
A: The property of material compatible with living tissue is called biocompatibility. Biocompatible…
Q: 16 C Fatty acyl CoA + 7 FAD + 7 NAD+ + 7 COA → 8 acetyl CoA + 7 FADH₂ + 7 NADH + 7 H* This is the…
A: Fatty acids are an important source of energy that are utilised for energy generation at the time of…
Q: Each turn of the citric acid cycle produces: * Oa) one molecule of ATP Ob) one molecule of FADH2 Oc)…
A: Introduction: Citric acid cycle is an aerobic process in which the breakdown of glucose molecule…
Step by step
Solved in 2 steps

- Define the following terms:a. prenylationb. steroidc. digitalisd. lipoproteine. apolipoproteinYou can choose one or more than one option Arginase is an enzyme that: is expressed in the cytoplasm of hepatocytes shows two isoenzymes in mammals. found in the liver and other tissues catalyzes the formation of urea and ornithine from arginine. induces hyperammonemia when it is overexpressed is inhibited by basic amino acids such as lysine and ornithine.You can choose one or more than one options Arginase is an enzyme that : BIOCHEMISTRY advanced is expressed in the cytoplasm of hepatocytes shows two isoenzymes in mammals. found in the liver and other tissues catalyzes the formation of urea and ornithine from arginine. induces hyperammonemia when it is overexpressed is inhibited by basic amino acids such as lysine and ornithine.
- The distal histidine stabilises the iron in heme group of a deoxyhaemoglobin. T/FWhen taken in appropriate amounts, vitamin E protectsthe body from ROS. However, when it is taken in excessiveamounts vitamin E can potentially make the body moresusceptible to ROS. Explain.A fixed amount of radiolabelled insulin was incubated with a fixed amount of anti-insulin antibody, the bound ligand was separated from free ligand at different time intervals. Draw a diagram of the concentration of the insulin/antibody complex with time. (Insulin/antibody complex is the bound ligand) asap typed only .
- Coenzyme Q-10 (Ubiquinone) is one of the most important and crucial substances within the human body. The heart muscle normally has very high levels of CoQ-10, and when these levels drop by 75%, it will stop pumping. When CoQ-10 (Ubiquinone) levels drop by 25% within the body, diseases usually occur. Why does this happen?A female neonate did well until approximately 24 hours of age when she became lethargic. A sepsis workup proved negative. At 56 hours, she started showing focal seizure activity. The plasma ammonia level was found to be 1,100 μmol/L (normal 5–35 μmol/L). Quantitative plasma amino acid levels revealed a marked elevation of argininosuccinate.19.3 Which one of the following enzymic activities is most likely to be deficient in this patient? EXPLAIN your answerA. Arginase.B. Argininosuccinate lyase.C. Argininosuccinate synthase.D. Carbamoyl phosphate synthetase I.E. Ornithine transcarbamoylase.A mutation in the alpha subunit of the G protein prevents it from binding to the adenylyl cyclase but not the recpetor. The amount of cAMP in cells will 1. stay the same 2. decrease 3. increase
- A defect in which of the following enzymes leads to Tay- Sachs disease? O Phospholipase CO a-galactosidase A O Hexosamidase A O SphingomyelinaseHDL-R: HDLs carry cholesterol to the liver for elimination. Liver cells respond to estrogen to express a protein receptor for HDLs coded by the HDL-R gene (HDL-R gene contains an estrogen response element.) PFK: codes for an enzyme called phosphofructokinase that is used in the first steps of breaking down glucose for energy in all cells. (Gene has no estrogen response element.) CD3: codes for a cell surface protein produced by immune B cells (Gene has no estrogen response element)mRNA transcripts can be collected from cells, made to glow with a fluorescent dye, and added to a microarray chip containing spots with DNA sequences from each of these genes. (In the depiction of the microarrays below ○ indicates no binding of the mRNA, and ● indicates binding.)Which of the gene expression patterns would you expect to see if you used mRNA from immune B cells to add to the microarray?What qualitative effect would you expect each of the following to have onthe P50 of hemoglobin?(a) Increase in pH from 7.2 to 7.4(b) Increase in PCO2 from 20 to 40 mm Hg(c) Dissociation into monomer polypeptide chains(d) Decrease in 2,3-BPG concentration from 7 mM to 5 mM in red cells.



