The instructor reveals that the weak acid is hypochlorous acid, HOCl. The known Ka for HOCl is 4.0×10-8. What is the percent error in the experiment?
Q: The calibration curve was generated using known concentrations of five solutions of a newly…
A: Introduction A calibration curve, sometimes referred to as a standard curve, is a common technique…
Q: Which of the following is incorrect: General steps in qualitative analysis include: Method…
A: A multiple choice question based on analytical methods, which is to be accomplished.
Q: The accuracy of an instrument was tested by conducting a Reference Standard Material (RSM)…
A:
Q: The molarity of a solution is determined by four separate titrations or replicates, where the…
A: Given observations are : 1). 0.4120 2). 0.4920 3). 0.3920 4). 0.4888 5). 0.4320 Standard error…
Q: Consider the structure of testosterone to identify the best suitable quantitative analysis for…
A: The steroid hormone testosterone is very significant. Total testosterone levels in healthy,…
Q: On their second titration trial, the CHM 111 student recorded an Initial titrant volume of 9.95 mL…
A: Given : Initial titrant volume = 9.95 mL And final titrant volume = 29.80 mL
Q: In this experiment it takes about 10 microliters of solution to produce a spot 1 cm in diameter. If…
A:
Q: The calibration curve was generated using known concentrations of five solutions of a newly…
A: The amount of a substance dissolved in a specified volume of the solvent produces a solution of a…
Q: 0.119 (measured) 3) Molarity of final unknown solution (calculated from calibration curve) M.
A: The slope-intercept form of line is y = mx + c, here, m = slope of the line and c = y-intercept. The…
Q: 3.) For part A.l in the experiment (see the lab data sheet), the identity of the acids were…
A: given values : H2SO4 - 3MHCl -6MCH3COOH - 6MH3PO4 Magnesium is added to 4 test tubes containing 4…
Q: AICI3 cl) OZN-Hg, HCI
A: The first step is Friedel craft acylation. In this step, benzene reacts with acyl ion. The second…
Q: 6. Given that the concentration of ascorbic acid is ~o.001 M in lemon juice, is this error…
A: Significant and Non significant error plays an important role in determining the accuracy of any…
Q: What do you call the type of analytical method that relies on the absolute stoichiometric…
A: Oxidative method - It a method involving transfer of electrons from oxidizing agent to the substance…
Q: The following volumes of 0.000300 M SCN- are diluted to 15.00 mL. Determine the concentration of…
A: Concentration of diluted SCN-1 can be calculated by using formulaM1V1 = M2V2Where M1 & V1 is the…
Q: Arrange the steps of gravimetric analysis in chronological order
A: Gravimetric analysis is a quantitative method of the measurement of the substance on basis of their…
Q: What is the importance of calibrating analytical balances?
A: Calibration of analytical balance is important.
Q: A group of students performed a series of titrations in the laboratory and found that their unknown…
A: 1. Given that, the exact value of concentration is Cexact = 15.47 M. The calculated value of…
Q: A volume of 25.00 mL of the original solution was diluted to a total volume of 100.0 mL and then…
A: If we take a definite amount of solution and then dilute it with by adding of water the mole of…
Q: You determine the acetic acid (HOAc) content of vinegar by titrating with a sodium hydroxide…
A: Step by step Explanation- The average of molar concentration is calculated as shown below M(NaOH)=…
Q: For EACH type of gravimetric analysis: Give actual applications of each analysis. Include the…
A: Gravimetric analysis: It is a quantitative analytical method that is based upon the measurement of…
Q: You have finished two trials of the sample analysis for the experiment and you noticed that the…
A: There are some tips and techniques which should be taken in mind while performing a titration.…
Q: Which of the following is incorrect: a. General steps in qualitative analysis include: Method…
A: Chemical analysis are of two types : 1. Qualitative analysis 2. Quantitative analysis The incorrect…
Q: Eight replicate measurements were made on a test solution producing the following results for the…
A:
Q: The analysis shows different concentrations based on replicate determinations of Zn in a water…
A: Absolute error is the difference between the measured value and actual value. Its modulus i.e.…
Q: Part B - Determination of Mg(OH)2 in Milk of Magnesia Mass of sample 50g Total Volume HCI used 80ml…
A: Total moles of HCl used can be given by using the following formula. Number of…
Q: what is the significance of gravimetry analysis?
A:
Q: could you please explain theses datas and graph for the synthesis and spectrophotemetric analysis of…
A: Interpretation: To explain the data of the graph and and spectrophotometric analysis of aspirin.
Q: Which of the following is incorrect: General steps in qualitative analysis include: Method…
A: Chemical analysis can be of two types, qualitative analysis, and quantitative analysis. Qualitative…
Q: Will Ag2 C03 (Ksp = 8.1 250.0ml of 0.0062 m 0.00014 m Na2 Co3 are
A: Solubility equilibrium of Ag2CO3 Ag2CO3(s) ----> 2Ag+(aq.) + CO32- Qsp = [Ag+]2[CO32-] =…
Q: The color change of a chemical requires an overtitration of 0.04 mL. Calculate the percent relative…
A: Given, Experimental value = 50 mL + 0.04 mL = 50.04 mL True value = 50 mL Calculating the percent…
Q: A student was given a stock ascorbic acid solution of 2.323 mg/L. Following the directions in the…
A: Concentration of stock ascorbic acid solution = 2.323 mg/L
Q: by 3. Nicotine is a weak base samples of a nicotine solution are titrated with 0.00526 M…
A: The question is based on the concept of titrations. we are titrating a nicotinic acid ( weak base)…
Q: Distinguish between: a. analysis (analyze) & determination (determine) b. qualitative & quantitative…
A: Distinguish between: a. analysis (analyze) & determination (determine)b. qualitative &…
Q: Starting from the primary standard solution in Question 4 and using the dilution scheme in Figure 1,…
A: The amount of solute that is dissolved in a specified volume of a solvent yields a solution. The…
Q: Based on the graph below, 1. molar concentration of HCO3- 2. alkalinity in ppm CaCO3
A: Step1: Here, the HCl is added to CaCO3. So, the reaction will be shown as Here, CaCO3 and HCl react…
Q: In a Spec-20 experiment a student took 4.3178g of an unknown material containing cobalt and…
A: Given data,Mass of sample=4.3178gMolarity of Co solution=0.075MVolume of solution=250mL=0.250L
Q: Write a step-by-step procedure for this serial dilution. The stock concentration of the blue dye…
A:
Q: H3CsH5O7 (aq) + 3 NaOH (aq) Na;C6H507(aq) + H2O (aq) Citric Acid Trial 1 Trial 2 Show Calculations…
A: The question is based on the concept of titrations. we are performing titration of citric acid with…
Q: Jake carried out three titrations to determine the concentration of unknown nitric acid (HNO3) using…
A: [NaOH] = 0.1050 M Vol of HNO3 used = 100.0 ml Vol of Na OH used (Va)= 20.56 ml Vol of Na OH used…
Q: 1. A student performed this experiment and obtained the following concentration values: 0.02813 M,…
A: As per our guidelines we can only solve first question. Please resubmit other questions as next…
Q: What is the absolute uncertainty and relative uncertainty of the molarity of the solution? M =…
A:
Q: A company illegally dumped their wastewater on a river bank. You are part of an environmental NGO…
A: As per our guidelines we should answer only one question. So kindly repost other questions as…
Q: The total hardness is due to one or a combination of Ca2+, Mg2+, and Fe2+ in your sample. It is…
A: According to Bartleby guidelines expert have to solve only one.
Q: Which of the following is incorrect: General steps in qualitative analysis include: Method…
A: Chemical analysis are of two types : 1. Quantitative analysis 2. Qualitative analysis The incorrect…
Q: During Lab 4, you are titrating a sample of new orange juice to determine its ascorbic acid (aka…
A: Amount of ascorbic acid can be calculated using proper dilution factors.
Q: A commercial cola brand states that the cola contains 45 mg phosphorus per 250 mL serving. Calculate…
A:
Q: Which of the following is incorrect: a. General steps in qualitative analysis include: Method…
A: A multiple choice question about chemical analysis, which is to be accomplished.
Q: Data ) Mass of Sodium Salicylate (g) 115a X 2) Concentration of Sodium Salicylate (M) 287 10M…
A: The concentration of the solution can be measured in various units the commonly used is molarity.…
Q: 4. In this experiment it takes about 10 microliters of solution to produce a spot 1 cm in diameter.…
A:
The instructor reveals that the weak acid is hypochlorous acid, HOCl. The known Ka for HOCl is 4.0×10-8. What is the percent error in the experiment?
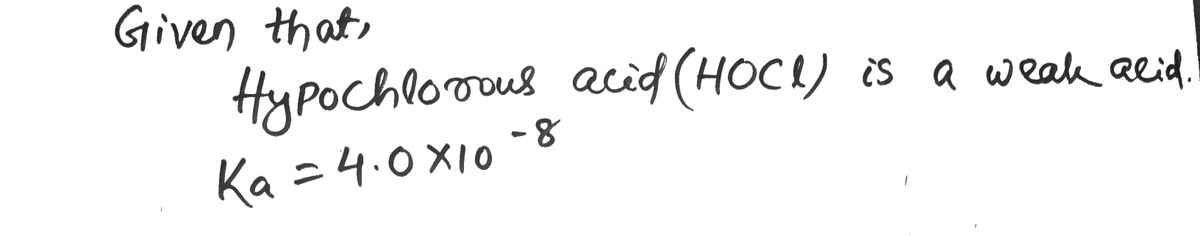
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- An excess of 0.34 mg Pb is obtained from an assay test determined in an analysis. What is the percent relative error in ppt if the mass of the sample is 550 g?The color change of a chemical requires an overtitration of 0.04 mL. Calculate the percent relative error if the total volume of the titrant is 50 mL.What are the types of gravimetric methods? Give an examples where each method is used in an analysis.
- What do you call the type of analytical method that relies on the absolute stoichiometric equivalences of balanced chemical reactions in order to compute the amount of analyte present in the sample? a.Oxidative methods b.Classical methods c. Blank Correction Methods d. Instrumental methodsWhat is the importance of calibrating analytical balances?Given that the concentration of ascorbic acid is sim0.001 Mi in lemon juice, is this error significant? Why or why not?
- The analysis shows different concentrations based on replicate determinations of Zn in a water sample: 0.754 mg, 0.721 mg, 0.734 mg, 0.731 mg and 0.742 mg. What is the absolute error if the standard solution has 0.750 mg of Zn?what is the significance of gravimetry analysis?For EACH type of gravimetric analysis:Give actual applications of each analysis. Include the SPECIFIC methodology for each sample chosen.
- You have finished two trials of the sample analysis for the experiment and you noticed that the burette is almost empty (~5 mL remaining). Should you add more titrant before the next sample analysis or not? Why or why not? Select one: YES. It will not affect the calculations and not cause error NO. It will affect the calculations and cause error Neither A nor B Cannot be determined1. A student performed this experiment and obtained the following concentration values: 0.02813 M, 0.02802, and 0.02788 M a. what is the mean concentration? b. what is the standard deviation of these results? 3. how would the following errors affect the concentration of Cl- obtained in question 2b? give your reasoning in each case. a. the student read the molarity of AgNo3 as 0.02104M instead of 0.02014M b. the student was past the endpoint of titration when he took the final buret reading.Consider the structure of testosterone to identify the best suitable quantitative analysis for testosterone?

