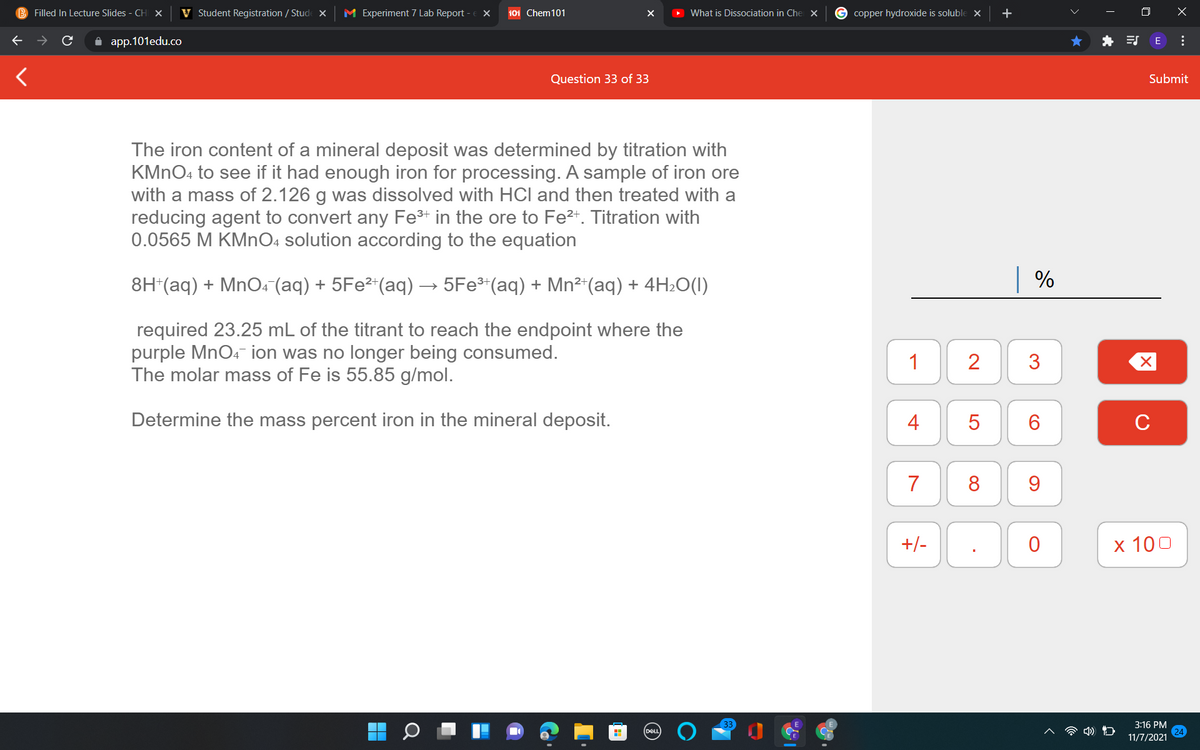
The iron content of a mineral deposit was determined by titration with KMNO4 to see if it had enough iron for processing. A sample of iron ore with a mass of 2.126 g was dissolved with HCl and then treated with a reducing agent to convert any Fe³+ in the ore to Fe²+. Titration with 0.0565 M KMNO4 solution according to the equation 8H*(aq) + MnO. (aq) + 5Fe²*(aq) → 5Fe*(aq) + Mn²*(aq) + 4H2O(I) required 23.25 mL of the titrant to reach the endpoint where the purple MnO4- ion was no longer being consumed. The molar mass of Fe is 55.85 g/mol. Determine the mass percent iron in the mineral deposit.
The iron content of a mineral deposit was determined by titration with KMNO4 to see if it had enough iron for processing. A sample of iron ore with a mass of 2.126 g was dissolved with HCl and then treated with a reducing agent to convert any Fe³+ in the ore to Fe²+. Titration with 0.0565 M KMNO4 solution according to the equation 8H*(aq) + MnO. (aq) + 5Fe²*(aq) → 5Fe*(aq) + Mn²*(aq) + 4H2O(I) required 23.25 mL of the titrant to reach the endpoint where the purple MnO4- ion was no longer being consumed. The molar mass of Fe is 55.85 g/mol. Determine the mass percent iron in the mineral deposit.
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter19: Transition Metals And Coordination Chemistry
Section: Chapter Questions
Problem 11E: Iron (II) can be oxidized to iron (III) by dichromate ion, which is reduced to chromium (III) in...
Related questions
Question
Transcribed Image Text:B Filled In Lecture Slides - CH
V Student Registration / Stude x
M Experiment 7 Lab Report
101 Chem101
> What is Dissociation in Che X
G copper hydroxide is soluble ×
+
A app.101edu.co
* E E
Question 33 of 33
Submit
The iron content of a mineral deposit was determined by titration with
KMNO4 to see if it had enough iron for processing. A sample of iron ore
with a mass of 2.126 g was dissolved with HCl and then treated with a
reducing agent to convert any Fe+ in the ore to Fe²*. Titration with
0.0565 M KMNO4 solution according to the equation
8H*(aq) + MnO4 (aq) + 5Fe²*(aq) → 5Fe³*(aq) + Mn²“(aq) + 4H2O(I)
| %
required 23.25 mL of the titrant to reach the endpoint where the
purple MnO4 ion was no longer being consumed.
The molar mass of Fe is 55.85 g/mol.
1
2
Determine the mass percent iron in the mineral deposit.
4
C
7
8
9.
+/-
х 100
3:16 PM
DEL
11/7/2021
...
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning