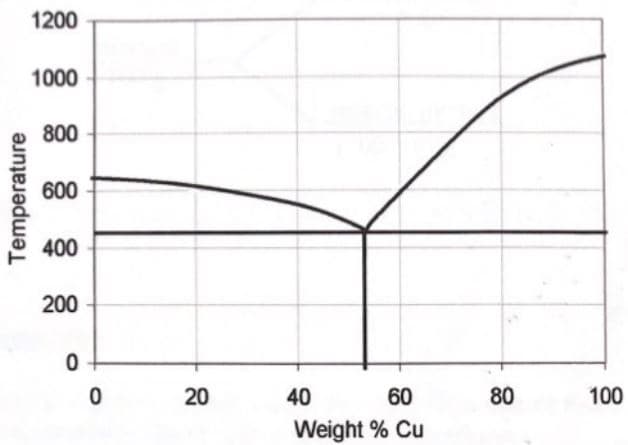
The phase diagram for the simple system aluminum copper is drawn below. Answer the following questions by referring to this diagram. A. How many phases will be present if an aluminum-copper mixture containing 80% copper is heated to 1000oC? B. If the system described in (A) is slowly cooled, at what temperature will a solid start to appear? What is this solid? C. What phases will be present if the mixture (A) is further cooled down to 600oC? D. What is the concentration of the saturated solution of copper in aluminum at 800oC? E. For a mixture given in (A), at what temperature will the eutectic mixture crystallize out? F. What is the percentage by weight of copper in the eutectic mixture formed in (E)?
3. The phase diagram for the simple system aluminum copper is drawn
below. Answer the following questions by referring to this diagram.
A. How many phases will be present if an aluminum-copper mixture containing 80% copper is heated to 1000oC?
B. If the system described in (A) is slowly cooled, at what temperature will
a solid start to appear? What is this solid?
C. What phases will be present if the mixture (A) is further cooled down to
600oC?
D. What is the concentration of the saturated solution of copper in
aluminum at 800oC?
E. For a mixture given in (A), at what temperature will the eutectic mixture
crystallize out?
F. What is the percentage by weight of copper in the eutectic mixture formed in (E)?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images









