The photo dimerization of benzophenone to benzopinacol is initiated by what type of electronic transition that then rapidly decomposes to a diradical since putting in electrons in anti bonds breaks bonds! The diradical then starts abstracting hydrogens from solution as pictured in the text? σ = electrons in sigma bonds n = electrons in non-bonding orbitals π electrons in pi bonds. anti bonds Hint: This process is very important because although the molecule responsible for human vision (retinal) is not a very long conjugated pi system, this transition allows retinal to absorb visible light. a. n to σ* b. σ to σ* C. π το π d. n to П e. σ to П* f. π to σ'
The photo dimerization of benzophenone to benzopinacol is initiated by what type of electronic transition that then rapidly decomposes to a diradical since putting in electrons in anti bonds breaks bonds! The diradical then starts abstracting hydrogens from solution as pictured in the text? σ = electrons in sigma bonds n = electrons in non-bonding orbitals π electrons in pi bonds. anti bonds Hint: This process is very important because although the molecule responsible for human vision (retinal) is not a very long conjugated pi system, this transition allows retinal to absorb visible light. a. n to σ* b. σ to σ* C. π το π d. n to П e. σ to П* f. π to σ'
Chapter3: Mechanisms
Section: Chapter Questions
Problem 105EQ
Related questions
Question
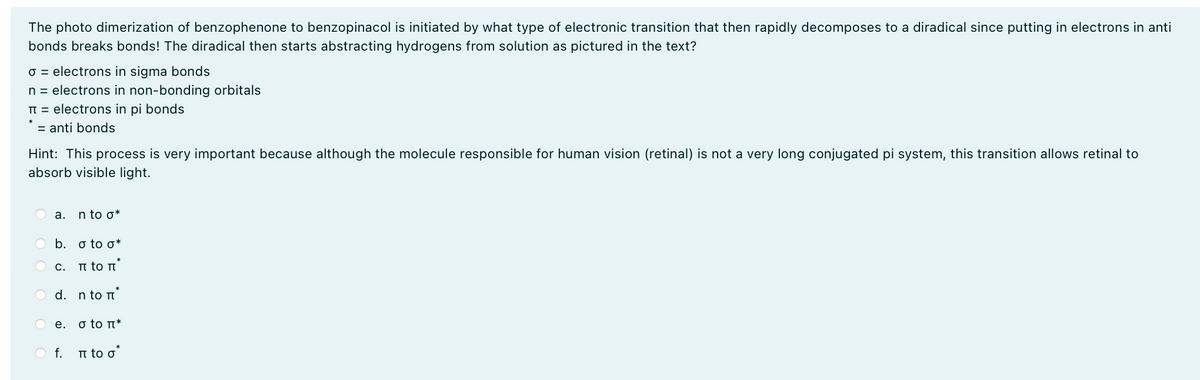
Transcribed Image Text:The photo dimerization of benzophenone to benzopinacol is initiated by what type of electronic transition that then rapidly decomposes to a diradical since putting in electrons in anti
bonds breaks bonds! The diradical then starts abstracting hydrogens from solution as pictured in the text?
σ = electrons in sigma bonds
n = electrons in non-bonding orbitals
π
electrons in pi bonds.
anti bonds
Hint: This process is very important because although the molecule responsible for human vision (retinal) is not a very long conjugated pi system, this transition allows retinal to
absorb visible light.
a.
n to σ*
b. σ to σ*
C.
π το π
d. n to П
e.
σ to П*
f. π to σ'
AI-Generated Solution
Unlock instant AI solutions
Tap the button
to generate a solution
Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole