The solubility of an unknown substance was tested three times during an experiment. Based on the solubility curve shown and the results of the experiment, what is most likely the identity of the substance? Select one: O sodium nitrate о potassium chloride O potassium nitrate O sodium chloride Solubility (grams of solute/100 g H₂0) 120 110 100 90 80 70 60 50 40 30 20 10 NaNO₂ SONY KCI NaCl 10 20 30 40 50 60 70 80 90 100 Temperature (°C) Mass of Unknown Solute That Dissolves in 100 g Water 50°C Trial 1 2 40°C 63.2 g 80.2 g 30°C 46.0 g 45.9 g 44.3 g 45.4 9 81.3 g 61.4 g 62.9 g 62.5 g 3 Average 79.8 g 80.4 g
The solubility of an unknown substance was tested three times during an experiment. Based on the solubility curve shown and the results of the experiment, what is most likely the identity of the substance? Select one: O sodium nitrate о potassium chloride O potassium nitrate O sodium chloride Solubility (grams of solute/100 g H₂0) 120 110 100 90 80 70 60 50 40 30 20 10 NaNO₂ SONY KCI NaCl 10 20 30 40 50 60 70 80 90 100 Temperature (°C) Mass of Unknown Solute That Dissolves in 100 g Water 50°C Trial 1 2 40°C 63.2 g 80.2 g 30°C 46.0 g 45.9 g 44.3 g 45.4 9 81.3 g 61.4 g 62.9 g 62.5 g 3 Average 79.8 g 80.4 g
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 66AP: Relative solubilities of salts in liquid ammonia can differsignificantly from those in water. Thus,...
Related questions
Question
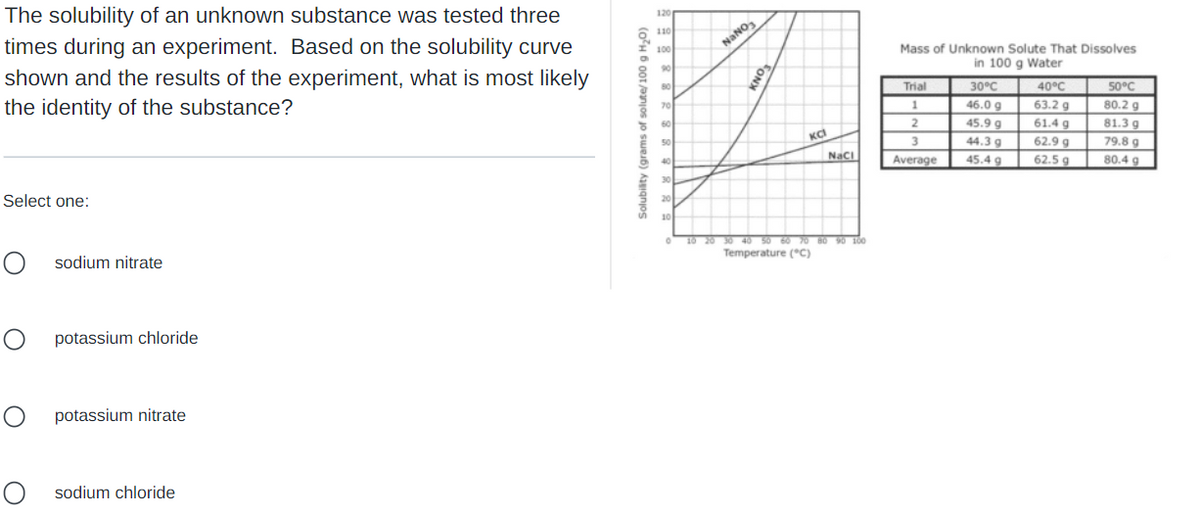
Transcribed Image Text:The solubility of an unknown substance was tested three
times during an experiment. Based on the solubility curve
shown and the results of the experiment, what is most likely
the identity of the substance?
Select one:
O sodium nitrate
о
potassium chloride
O
potassium nitrate
Osodium chloride
Solubility (grams of solute/100 g H₂O)
120
110
100
90
80
70
60
50
40
30
20
10
0
NaNO3
KNO3
ка
NaCl
10 20 30 40 50 60 70 80 90 100
Temperature (°C)
Mass of Unknown Solute That Dissolves
in 100 g Water
Trial
1
2
3
Average
50°C
80.2 g
30°C
46.0 g
45.9 g
44.3 g
45.4 g
40°C
63.2 g
61.4 g
81.3 g
79.8 g
62.9 g
62.5 g
80.4 g
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
