The solubility of silver nitrate has a [Blank] relationship with the temperature of the water. The solubility of silver nitrate in water can be described as [Blank] At [Blank] ℃ exactly 216 grams of silver nitrate should make a saturated solution. At 60 ℃, it should take [Blank] grams of silver nitrate to make a saturated solution. After studying the table, is it reasonable to say that silver nitrate dissolves better at higher temperatures? Blank 1 options- Positive, Negative, Neutral Blank 2 options- Insoluble, Slightly soluble, Highly Soluble Blank 3 options- 0, 20,40,60,80 Blank 4 options- 122, 215, 311, 440, 585 Blank 5 options- Yes, No
The solubility of silver nitrate has a [Blank] relationship with the temperature of the water. The solubility of silver nitrate in water can be described as [Blank] At [Blank] ℃ exactly 216 grams of silver nitrate should make a saturated solution. At 60 ℃, it should take [Blank] grams of silver nitrate to make a saturated solution. After studying the table, is it reasonable to say that silver nitrate dissolves better at higher temperatures? Blank 1 options- Positive, Negative, Neutral Blank 2 options- Insoluble, Slightly soluble, Highly Soluble Blank 3 options- 0, 20,40,60,80 Blank 4 options- 122, 215, 311, 440, 585 Blank 5 options- Yes, No
Chemistry: Matter and Change
1st Edition
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Chapter14: Mixtures And Solutions
Section: Chapter Questions
Problem 114A
Related questions
Question
100%
The solubility of silver nitrate has a [Blank] relationship with the temperature of the water.
The solubility of silver nitrate in water can be described as [Blank]
At [Blank] ℃ exactly 216 grams of silver nitrate should make a saturated solution.
At 60 ℃, it should take [Blank] grams of silver nitrate to make a saturated solution.
After studying the table, is it reasonable to say that silver nitrate dissolves better at higher temperatures?
Blank 1 options- Positive, Negative, Neutral
Blank 2 options- Insoluble, Slightly soluble, Highly Soluble
Blank 3 options- 0, 20,40,60,80
Blank 4 options- 122, 215, 311, 440, 585
Blank 5 options- Yes, No
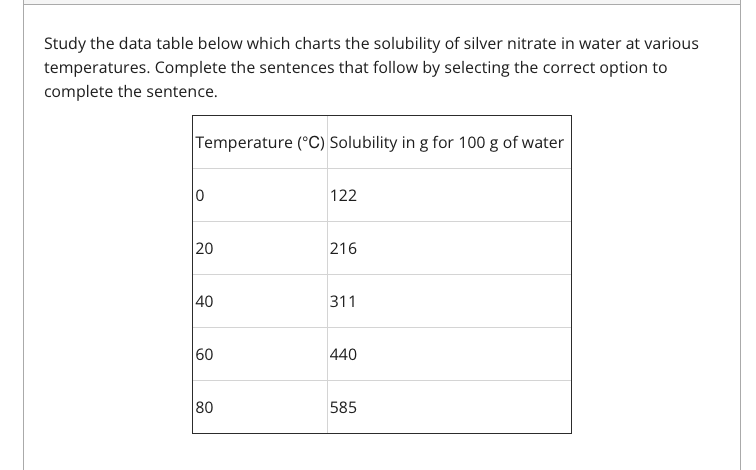
Transcribed Image Text:Study the data table below which charts the solubility of silver nitrate in water at various
temperatures. Complete the sentences that follow by selecting the correct option to
complete the sentence.
Temperature (°C) Solubility in g for 100 g of water
122
20
216
40
311
60
440
80
585
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning