The voltaic cell shown above has two identical zinc electrodes : The solution in half-cell A contains a 4.2 mol/L ZnSO4 solution. Half-cell B solution contains a 0.043 mol/L ZnSO4 solution. Calculate the emf (electromotive force) of this cell at non-standard conditions in volts (V)
The voltaic cell shown above has two identical zinc electrodes : The solution in half-cell A contains a 4.2 mol/L ZnSO4 solution. Half-cell B solution contains a 0.043 mol/L ZnSO4 solution. Calculate the emf (electromotive force) of this cell at non-standard conditions in volts (V)
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter25: Nucleotides, Nucleic Acids, And Heredity
Section: Chapter Questions
Problem 25.41P
Related questions
Question
100%
The voltaic cell shown above has two identical zinc electrodes :
The solution in half-cell A contains a 4.2 mol/L ZnSO4 solution.
Half-cell B solution contains a 0.043 mol/L ZnSO4 solution.
Calculate the emf (electromotive force) of this cell at non-standard conditions in volts (V).
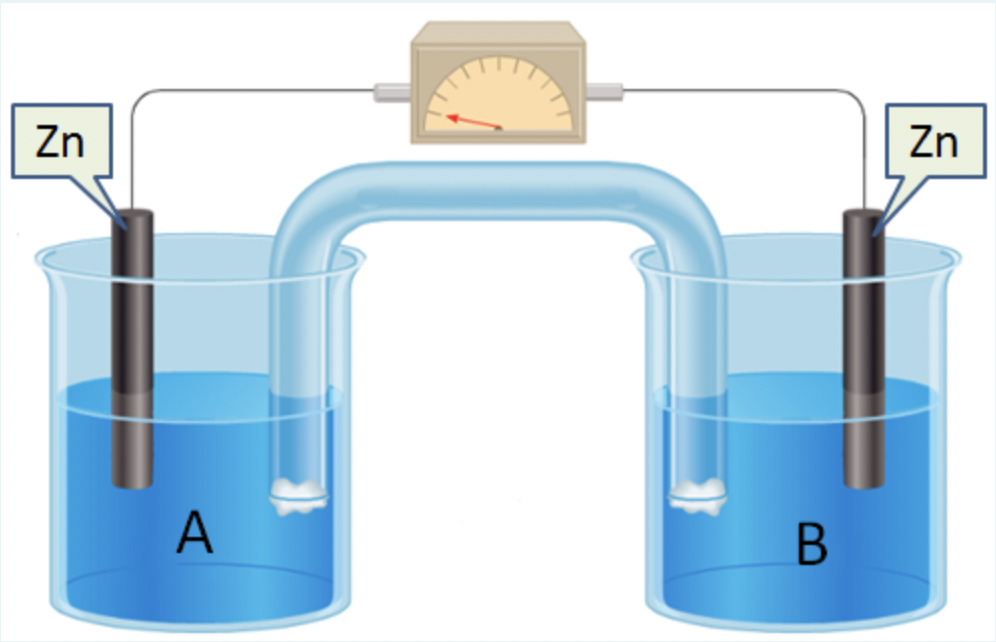
Transcribed Image Text:Zn
Zn
A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning
