True False Granules may be swallowed, dispersed in food, or dissolved in water. Granules are usually less stable to the effects of atmospheric humidity. An effervescent dosage form is dissolved or dispersed in water to initiate the effervescence prior to ingestion
True False Granules may be swallowed, dispersed in food, or dissolved in water. Granules are usually less stable to the effects of atmospheric humidity. An effervescent dosage form is dissolved or dispersed in water to initiate the effervescence prior to ingestion
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter21: Structure And Bonding In Solids
Section: Chapter Questions
Problem 57AP
Related questions
Question
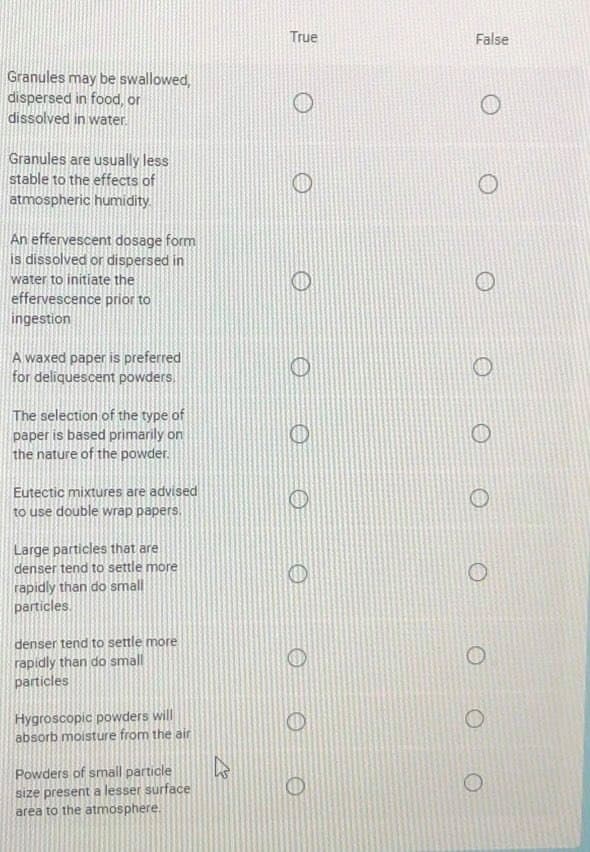
Transcribed Image Text:True
False
Granules may be swallowed,
dispersed in food, or
dissolved in water.
Granules are usually less
stable to the effects of
atmospheric humidity.
An effervescent dosage form
is dissolved or dispersed in
water to initiate the
effervescence prior to
ingestion
A waxed paper is preferred
for deliquescent powders.
The selection of the type of
paper is based primarily on
the nature of the powder.
Eutectic mixtures are advised
to use double wrap papers.
Large particles that are
denser tend to settle more
rapidly than do small
particles.
denser tene
to settle more
rapidly than do small
particles
Hygroscopic powders will
absorb moisture from the air
Powders of small particle
size present a lesser surface
area to the atmosphere.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning