TUTOR Predicting Relative lonization Energy 8A 1A H 2A 4A 5A 6A 7A He 3A Li Be BCNOF Ne Na Mg 38 4B 5B 6B 78 r 8B 1B 28 AI SIPS CI Ar K Ca Sc Tiv Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y 2r ND Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Fr Ra Ac Rf Ha Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa UNp Pu Am Cm Bk Cf Es Fm Md No Lr Using only the periodic table arrange the following elements in order of increasing ionization energy: polonium, oxygen, tellurium, sulfur Lowest Highest oxygen Xsulfur x tellurium x polonium Please answer this question according to the general rules you have learned regarding periodic trends. DO NOT base your answer on tabulated values since exceptions may occur. Resubmit Show Approach Show Tutor Steps Incorrect Previous 近
TUTOR Predicting Relative lonization Energy 8A 1A H 2A 4A 5A 6A 7A He 3A Li Be BCNOF Ne Na Mg 38 4B 5B 6B 78 r 8B 1B 28 AI SIPS CI Ar K Ca Sc Tiv Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr Rb Sr Y 2r ND Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe Cs Ba La Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn Fr Ra Ac Rf Ha Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa UNp Pu Am Cm Bk Cf Es Fm Md No Lr Using only the periodic table arrange the following elements in order of increasing ionization energy: polonium, oxygen, tellurium, sulfur Lowest Highest oxygen Xsulfur x tellurium x polonium Please answer this question according to the general rules you have learned regarding periodic trends. DO NOT base your answer on tabulated values since exceptions may occur. Resubmit Show Approach Show Tutor Steps Incorrect Previous 近
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter4: Introduction To Quantum Mechanics
Section: Chapter Questions
Problem 39P: Chapter 3 introduced the concept of a double bond between carbon atoms, represented by C=C , with a...
Related questions
Question
What is wrong with my answer?
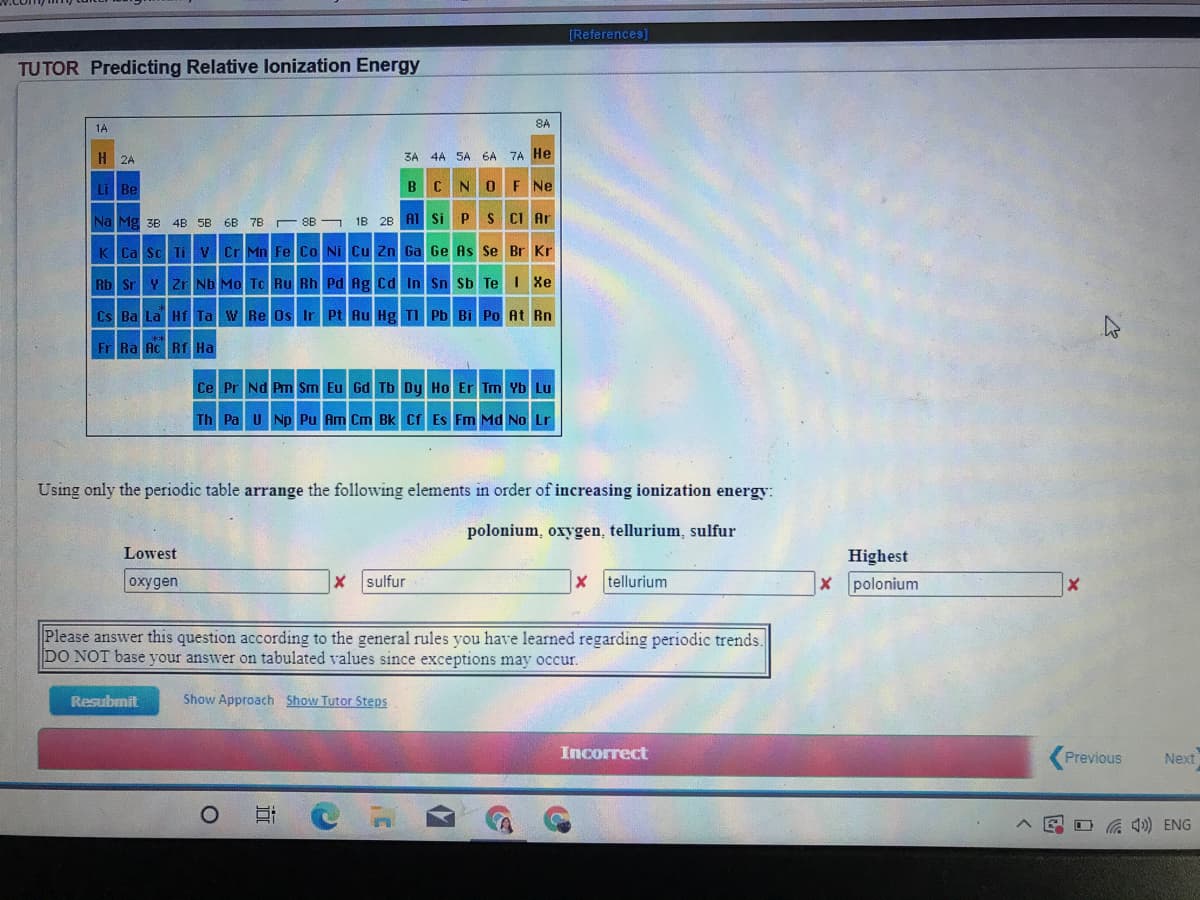
Transcribed Image Text:[References)
TUTOR Predicting Relative lonization Energy
8A
1A
H 2A
3A 4A 5A 6A 7A He
Li Be
BCNOF Ne
Na Mg 38 4B 5B
- 8B - 18 28 A1 Si P S CI Ar
6B 7B
K Ca Sc TiVCr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr
Rb Sry zr ND Mo TC Ru Rh Pd Ag Cd In Sn Sb Te IXe
Cs Ba La Hf Ta W Re Os Ir Pt Au Hg TI Pb Bi Po At Rn
Fr Ra Ac Rf Ha
Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
Th Pa U Np Pu Am Cm Bk Ccf Es Fm Md No Lr
Using only the periodic table arrange the following elements in order of increasing ionization energy:
polonium, oxygen, tellurium, sulfur
Lowest
Highest
oxygen
sulfur
x tellurium
X polonium
Please answer this question according to the general rules you have learned regarding periodic trends.
DO NOT base your answer on tabulated values since exceptions may occur.
Resubmit
Show Approach Show Tutor Steps
Incorrect
Previous
Next
4>) ENG
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
