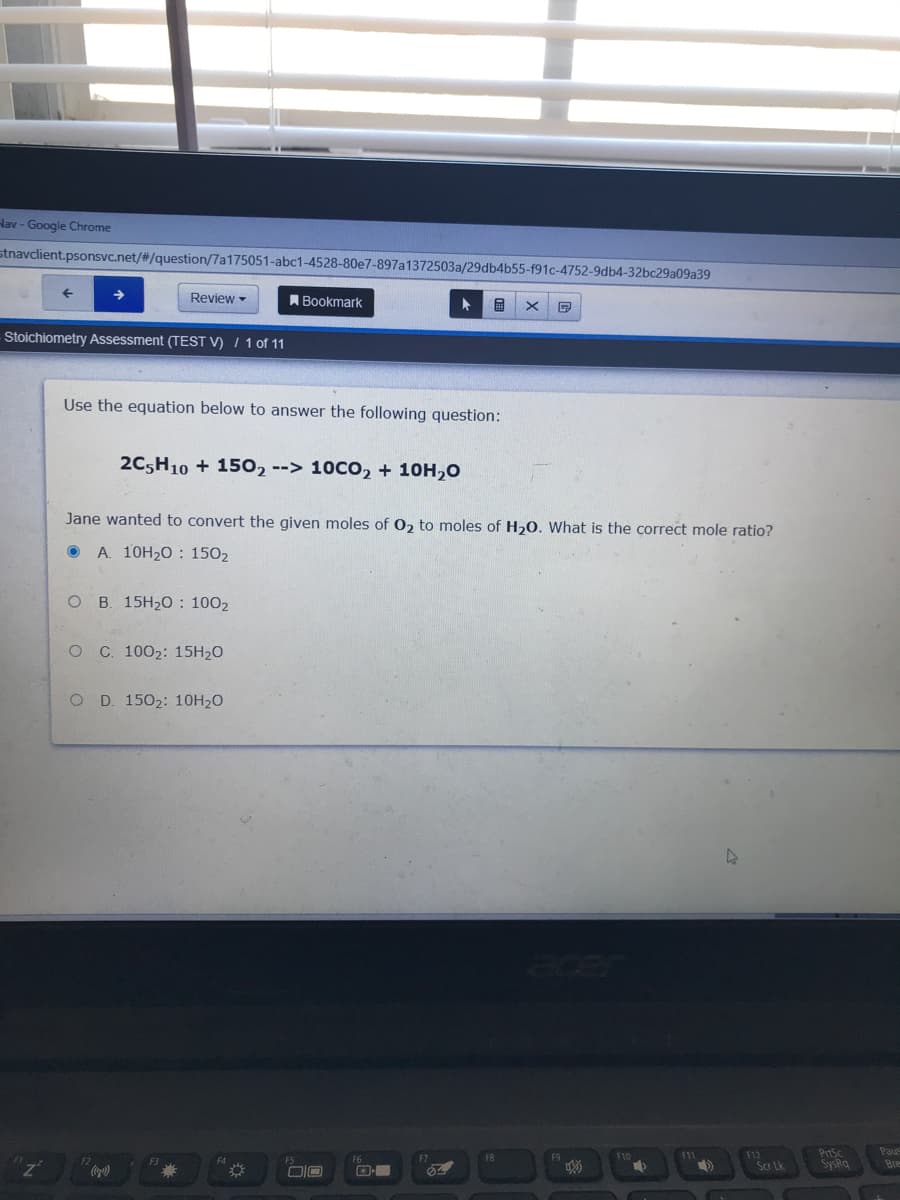
Use the equation below to answer the following question: 2C5H10 + 1502--> 10CO, + 10H,0 Jane wanted to convert the given moles of 02 to moles of H20. What is the correct mole ratio? A. 10H20 : 1502 ов 15H20: 1002 о с. 1002: 15H20 O D. 1502: 10H20
Use the equation below to answer the following question: 2C5H10 + 1502--> 10CO, + 10H,0 Jane wanted to convert the given moles of 02 to moles of H20. What is the correct mole ratio? A. 10H20 : 1502 ов 15H20: 1002 о с. 1002: 15H20 O D. 1502: 10H20
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
100%
Transcribed Image Text:Hav - Google Chrome
stnavclient.psonsvc.net/#/question/7a175051-abc1-4528-80e7-897a1372503a/29db4b55-f91c-4752-9db4-32bc29a09a39
Review -
A Bookmark
Stoichiometry Assessment (TEST V) / 1 of 11
Use the equation below to answer the following question:
2C5H10 + 1502--> 10CO2 + 10H,O
Jane wanted to convert the given moles of 0, to moles of H20. What is the correct mole ratio?
A. 10H20 : 1502
O B. 15H20 : 1002
O C 1002: 15H20
O D 1502: 10H20
PrtSc
SysRa
Paus
F12
Sa Lk
FB
F10
F11
Bre
Coca)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



