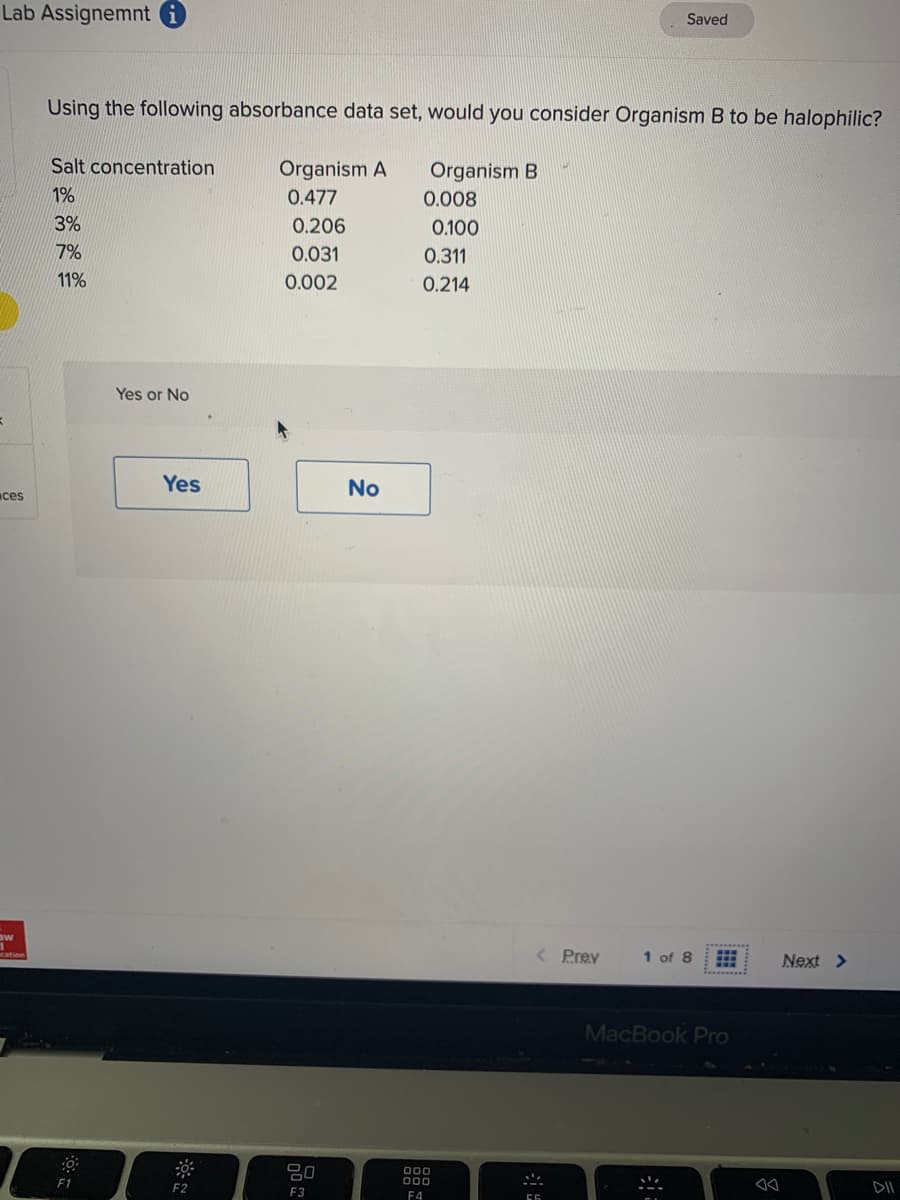
Using the following absorbance data set, would you c Salt concentration Organism A Organism B 1% 0.477 0.008 3% 0.206 0.100 7% 0.031 0.311 11% 0.002 0.214
Q: While measuring the turbidity of a bacterial culture in class, Kevin recorded both the absorbance…
A: Turbidity is the measurement of the suspended particles in a fluid that makes it opaque. Quality…
Q: explain the results of this experiment Table 1 Absorption Spectrum of Cobalt Chloride Using a…
A: We know that the calibration curve can be used to determine the concentration of an unknown…
Q: When calibrating a pH meter, 3 (three) standard color-coded buffer solutions are used, which of…
A: pH is a quantitative measure of how acidic or basic is the aqueous or other liquid solution is. It…
Q: You use a spectrophotometer to measure the absorbance of a colored solution and find it to be 1.6.…
A: According to Beer- Lambert law when a monochromatic light passes the homogeneous medium, it reflect,…
Q: in a solution, the solvent is: A) always a gas B) always water C) the substance that is…
A: A solution is a uniform and stable mixture of solute and solvent. Solute particles are invisible in…
Q: When a thermometer is placed in a substance, what is it actually measuring? density heat pressure…
A: Different instruments are used to measure volume, density, heat, pressure, and temperature, etc.…
Q: Which one of the following laws for dissolved gases in water is NOT true? O a Warm water can hold…
A: Water is the universal solvent. It is made up of two hydrogen and one oxygen molecule.
Q: BSA (mg/ml) Absorbancy 595nm 0 0.225 1 0.310 2 0.420 3 0.510 4 0.610 5 0.720 6 0.810 7 0.915 8 0.950…
A: The Bradford assay is quick and requires roughly the same quantity of protein as the Lowry assay.…
Q: How do I find the amount of substrate consumed using the given concentration of substrate, measured…
A: A standard curve, or calibration curve or calibration line, is a type of graph used in quantitative…
Q: . Which of the following correctly describe the colligative properties of aqueous solutions? I…
A: Biochemistry is an interdisciplinary field involving subjects such as biology and chemistry. It…
Q: RELATIVE HUMIDITY 1. the amount of water vapor in grams per cubic meter of air at a given…
A: Humidity is known as the presence of water vapor in the atmosphere. This is measured by an…
Q: Which water sample would dissolve the most oxygen? O Pure water at 21 C Seawater at 21°C Seawater at…
A: The solubility of oxygen in water is known as dissolved oxygen. It depends upon several factors such…
Q: The colligative properties depend on the concentration based on the solvent. True O False
A: The given statement is true . The colligative properties depend on the concentration based on the…
Q: Bastian created a dilution series with Blue #2 dye, similar to the one you will complete in Part C…
A: The concentration of a substance can be determined using spectrophotometry. The sample containing…
Q: Which of the following depicts the mild alkalosis? Select one: a. pH=7.33 Ob. pH=7.45 O c. H=7.65…
A: The collecting duct of kidney participates in reabsorption and secretion of selective ions into…
Q: How would I graph a scatterplot/ x-y plot of temperature and absorbance. Temperature is the…
A: Should plot a graph between temperature and absorbance using five parts of graph rule.
Q: When doing the glucose in drinks experiment I measured an absorbance reading of 0.002 for orange…
A: Light is made up of many colors, which are referred to as wavelengths . Each color has its own…
Q: Given a choice of inhaling NO or eating some oxide of N (e.g. nitrites). Which one would you rather…
A: Nitric oxide is defined as a molecule that is produced naturally by the body. It is important for…
Q: Why are quantitative absorbance measurements made at lambda max?
A: Introduction :- Using spectrum of wavelength , different levels of absorption can be calculated. It…
Q: | || | | || | || 1 45 Using the figure, which lane has a poor quality sample and is possibly…
A: Electrophoresis (Gel electrophoresis) is commonly used in labs to study a mixture of biomolecules…
Q: What would the dissolved oxygen level most likely be if the temperature of the water was 35C
A: Dissolved Oxygen and Water -- Introduction -- For drinking and other activities there must be a…
Q: Suppose an unknown sample is too concentrated (the absorbance was off the scale) Explain how you…
A: To determine the actual concentration of Bovine serum albumin in a highly concentrated unknown…
Q: Which among the following is an example of a pharmaceutically important buffer system? a. All of…
A: A buffer is a solution that can withstand pH changes when acidic or basic components are added. Many…
Q: Put these aqueous solutions in order of decreasing freezing point 0.1 m NaCl 0.1 m MgCl2 0.1 m…
A: For NaCl, i=2 as two ions Na+ and Cl- will be generated in aqueous solution MgCl2 i=3 as three ions…
Q: explain what the results of this experiment are and the purpose Table 2 Absorbance vs CoCl2…
A: The Beer-Lambert Rule: The concentration of the absorbing species and the route length of light…
Q: To achieve a hundred fold dilution you would add _____ microliters of sample to the appropriate…
A: The dilution method is used to achieve the desired concentration of the sample in a tube or a…
Q: Which mixture acts as buffer solutions? O 50 ml 0.1M H2CO3 and 100 ml 0.1M NAOH 100 ml 0.1M H2CO3…
A: A buffer solution is composed of a weak acid and its conjugate base. The buffer solution resists the…
Q: Suppose two water samples have the followin orms of nitrogen at day zero. Sample A Sample B (mg/L)…
A: BOD or Biochemical Oxygen demand gives the amount of oxygen taken by the microorganisms for…
Q: Explain why when non-polar molecules are placed in water, they cause the entropy of the system to…
A: Hydrophobes are nonpolar molecules and usually have a long chain of carbons that do not interact…
Q: From your data in Table 8.2 choose the correct pH for household chemical listed here. pH 6 purple pH…
A: The pH is considered as the scale, which is used to specify the basicity as well as the acidity of…
Q: Using absorbance readings and a standard curve relating absorbance to cell number, you suspect that…
A: The countable range of colonies for an ideal plate count method is considered 30-300 colonies per…
Q: Compute for the sodium concentration of the patient. Use the same formula in the computation of the…
A: Spectrophotometry measures the absorbance spectrum of a substance. It measures the absorbance…
Q: Below is a BSA standard curve and the duplicate values of absorbance for 3 samples. Calculate the…
A: Standard curve is made with known concentrations of Bovine serum albumin (BSA) to find the unknown…
Q: 2. What volume of 0.100 M HCI would have to be added to this buffering system, to reduce the pH to…
A: The pH of the acidic buffer of blood is given as:pH=pKa+log[bicarbonate][carbonic acid]
Q: You obtained the following raw data when setting up a Bradford standard curve: BSA (mg/ml)…
A: The Bradford Standard curve have BSA (Bovine Serum Albumin ) concentration in X-axis and Absorbance…
Q: A student conducted an experiment to see how the amount of dissolved salt affects the boiling point…
A: Various factors are involved in determining the boiling and melting point of a substance. It may…
Q: Measurement of Bacterial Growth in LB, pH 3.2 Plot of Absorbance vs. Time (mins) c 37c 45'c 0.6 0.4…
A: The bacterial growth curve consists of four distinct growth phases: Lag Phase, Exponential Phase,…
Q: Conc Abs 0.983 0.821 0.876 0.689 0.624 0.427 0.411 0.223 0.297 0.147 Construct a curve and using…
A: In the absorbance vs concentration graph let us keep the absorbance in the Y-Axis and concentration…
Q: 1. compute for patient B result of his chloride analysis with the following data using mg/dL unit…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: If I add 10 microliters of a culture at 106 cells/mL to 990 microliters of nutrient broth, what is…
A: Cell density refers to number of cells present per unit volume. It is usually expressed in cells/mL.
Q: a) proportional to the charges on the two interacting atoms. b) proportional to the dielectric…
A: Electrostatic interactions are found to be a strong factor that determines the conformation of…
Q: When a SBIA021 student measured the pH of 1.0M HCI, the student obtained a pH meter and failed to…
A: Either separate reference and pH electrodes or a single combination electrode are used in pH metres.…
Q: Which of the ff. is carried out when sample has a low absorbance? Use a cuvette with shorter width.…
A: There are various methods for quantitative estimations of solutions. The colorimeter and…
Q: Solution A is 20 degrees celsius, Solution B is 80 degrees celsius (both are the same kind of…
A: The amount of heat required per unit mass to increase the temperature by one degree Celsius is known…
Q: A team of investigators is out on a boat on a lake on a marvelous,sunny summer day, and they are…
A: Introduction Ocean have totally different ecosystem as there are different factors that affect the…
Q: When measuring the absorbance of methylene blue solution containing corn starch, how would corn…
A: Starch is a homopolysaccharide composed of glucose units. The starch which is present in and…
Q: How much heat would be required to convert 7.47 mol of a pure substance from a liquid at 40.0°C to a…
A: The heat is a form of energy. The heat of vaporization is defined as the amount of heat needed to…
Q: Suppose you have an initial chlorine concentration of 5%. Then out of that, you are told to prepare…
A: The concentration of a solution: The concentration of a solution is defined as the amount of the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- you did a serial dilution experiment to determine the CFU/table of the Equate Children's probiotics. Given the ingredient list shown, how many colonies would you expect to see on a plate if you plated 0.1 mL from the 10-7 dilution tube, and incubated aerobically at 37 ⁰C for several days?Guanosine (C10H13N5O5) in solution has a maximum absorbance at a wavelength of 275 nm. The molar extinction co-efficient at this wavelength is 84M−1cm−1and the path length is 24.7 cm. Through the use of a spectrophotometer, it is found that the that A275= 1.48. What is the concentration of the guanosine solution in grams/litre? Molecular weights (g/mol): C-12, H-1, N-14, O-16 Select one: A. 0.201869 g/L B. 0.435188 g/L C. 0.059919 g/L D. 0.294046 g/L E. 0.000713 g/La. Let’s say for example that a milk sample has 10,000 bacteria per milliliters. If 1 mL of this sample were plated out, these would theoretically be 10,000 colonies in the Petri plate. Discuss and explain the serial dilutions of this example. b. The disk diffusion method was used to evaluate 3 disinfectants. The results were as follows: Solution Zone of inhibition X 0 mm Y 5 mm Z 10 mm How would you interpret? Provide inference.
- You did a serial dilution experiment to determine the CFU/table of the Webbers' probiotics. Given the ingredient list shown, how many colonies would you expect to see on a plate if you plated 0.1 mL from the 10-6 dilution tube, and incubated aerobically at 37 ⁰C for several days?in food microbiology, how do you compute for concentration (M) and absorbance (A) of peroxidase activity on hydrogen peroxide at different pH values? (wavelength used = 415nm). can you please explain the calculations thank you if given: Temperature = 75 C Molar absorptivity coefficient = 10.5 Path length (cm) = 2 I = transmitted light = 0.45Suppose you inoculated two 0.4% semi-solid agar deeps: one deep was inoculated with a motile organism and the other deep with a non-motile organism. You (in a stroke of genius) found a way to quantitatively analyze the deeps using a spectrophotometer. Which deep would have the highest absorbance and why?
- You record the absorbance of your unknown BSA sample at 0.276. What is the concentration in ug/mL of the unknown assuming you have a standard curve with the line of best fit with y=0.0001x+0.0038 as its equationI do not understand the results of this experiment, explain what the results of this experiment are and the purpose Table 2 Absorbance vs CoCl2 concentration Test Tube Number Cobalt Chloride Concentration (mol/mL) Absorbance at 510 nm 1 0.000 0.000 2 0.009 0.024 3 0.018 0.055 4 0.027 0.085 5 6 0.036 0.045 0.112 0.138 7 (Unknown) 0.088If the volume of a Staphylococcus aureus cell is estimated at 0.5 μm3, how many cells could be accommodated, in principle, in 5 mL of saturated culture? (1 mL = 1 cm3). Show your calculations.
- a. Is there a microbe that could grow on a medium that contains onlythe following compounds dissolved in water: CaCO3, MgNO3,FeCl2, ZnSO4, and glucose? Defend your answer.b. A useful mnemonic device for keeping track of major elementsrequired by most organisms is: C HOPKINS Ca Fe, Na ClMgood. Read aloud, it becomes “See Hopkin’s Cafe, NaCl (salt)M good!” What does each letter stand for?How would I graph a scatterplot/ x-y plot of temperature and absorbance. Temperature is the independent variable (x-axis) and absorbance is the dependent variable(y-axis)FOV measured at 100x total magnification is 400 micrometers. What is the FOV at 40X total magnification? 2. If FOV is 5 mm and the number of cells in FOV is estimated at 35, what is the cell size in mm? In micrometers?



