Using the reagents available in Table 4.2 of the lab manual, which answer correctly represents all of the reagents Jason used for his Part B procedure in order to obtain the products shown in the particle diagram? O 3 M H3PO4 and then Mg metal O 3 M H2SO4 and then Mg metal O 3 M H2SO4 and then Zn metal O 3 M H2SO4 and then Cu metal O 3 M NAOH and then Cu metal
Using the reagents available in Table 4.2 of the lab manual, which answer correctly represents all of the reagents Jason used for his Part B procedure in order to obtain the products shown in the particle diagram? O 3 M H3PO4 and then Mg metal O 3 M H2SO4 and then Mg metal O 3 M H2SO4 and then Zn metal O 3 M H2SO4 and then Cu metal O 3 M NAOH and then Cu metal
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
ChapterL3: Carbon (13c) Nmr Spectroscopy
Section: Chapter Questions
Problem 13CTQ
Related questions
Question
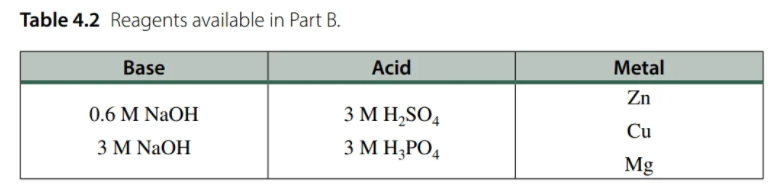
Transcribed Image Text:Table 4.2 Reagents available in Part B.
Base
Acid
Metal
Zn
0.6 M NaOH
3 M H,SO4
Cu
3 M NAOH
3 M H,PO4
Mg
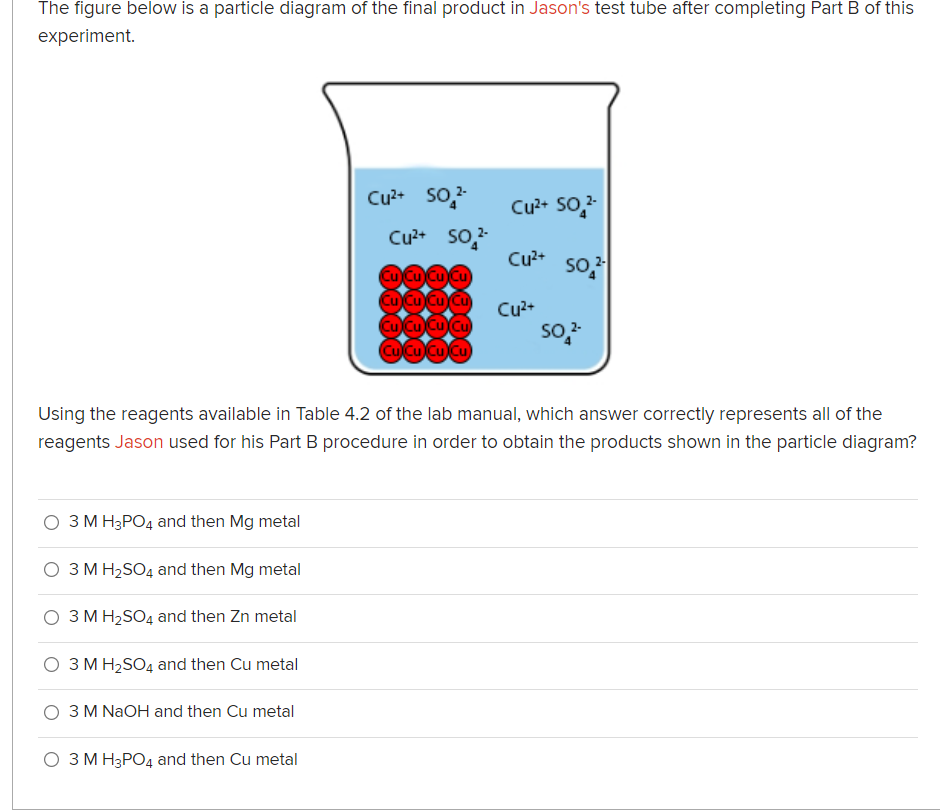
Transcribed Image Text:The figure below is a particle diagram of the final product in Jason's test tube after completing Part B of this
experiment.
Cu²+ so,-
Cu2+ So,
so
Cu²+
Cu²+
so,
Cu Cu Cu cu
Cu Cu Cu cu
Cu Cu cu Cu
Cu2+
so,
Using the reagents available in Table 4.2 of the lab manual, which answer correctly represents all of the
reagents Jason used for his Part B procedure in order to obtain the products shown in the particle diagram?
O 3 M H3PO4 and then Mg metal
O 3 M H2SO4 and then Mg metal
O 3 M H2SO4 and then Zn metal
O 3 M H2SO4 and then Cu metal
O 3 M NAOH and then Cu metal
O 3 M H3PO4 and then Cu metal
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning