Q: HC=CH The synthesis above can be performed with some combination of the reagents listed below. Give ...
A: For deprotonation of acetylene we need strong base such as NaNH2.
Q: The hydrolysis of a substance is specifically catalyzed by hydrogen ions, and the rate constant is g...
A:
Q: A laboratory stock solution is 1.50 M NaOH. Calculate the volume of this stock solution that would b...
A:
Q: 4) Rank the F/U in order of increasing pH. Explain the basis of your ranking. Assume the concentrati...
A: In order of rising pH, rank the F/U. The more electronegative an element is, the less stable its lon...
Q: What is the unit of absorbance? b)When he exponential trend line of iron concentration vs time i...
A: a) Absorbance has not unit. It is dimensionless.
Q: The rate constants for the decomposition of a hydrocarbon were measured at different temperatures as...
A:
Q: An electron on a hydrogen atom emits light at a wavelength of 434 nanometers when falling to the n =...
A: We know, The general formula for the hydrogen emission spectrum is given by- ν¯=R1n12-1n22 where ν¯ ...
Q: Which of the following will result to an unlit light bulb when subjected to the conductivity test? ...
A: A multiple choice question based on electrolysis that is to be accomplished.
Q: HO. Но
A: Organic reaction mechanisms:
Q: How many moles are in 3.01x10^23 particles? * O 1.00mol O 0.500mol O 18.1mol O 2.00mol
A: we know that 1mole contains 6.022 *10²³ particles.
Q: Acetone has a vapor pressure of 286.5 mm Hg at 25°C and a normal boiling point of 56.2°C. What is th...
A:
Q: The Molarity of the solution The molality of the solution The mole fraction of water
A:
Q: 5. Draw the Electron Structure for Be & N 6. Draw the Electron Structure Formula Unit: Formula Unit:...
A: The Formula Unit is = Be3N2 Calcium Phosphide structure The Formula Unit is = Ca3P2
Q: A chemical factory has been illegally disposing their chemical wastes without necessary pretreatment...
A: pH of a solution is defined as the negative logarithmic of hydrogen ion Concentration.
Q: 122 CHAPTER 5ID 15. Which of the following best describes the particle-level bonding in a crystal of...
A: The correct option are as follows:
Q: 1. Will Agl03 precipitate when 20 mL of 0.010 M AgIO3 is mixed with 10 mL of 0.015 M NalO3? Ep of Ag...
A: Solution -
Q: A sample consisting of 1 mol of dioxygen molecules with C, m= 29.4 JK 'mot! has an initial volume of...
A: Given Initial volume = 0.06 m3 Final volume = 0.02 m3 Initial temperature = 270 k Moles = 1 Cp, m =...
Q: Put the compounds drawn below in order of increasing basicity (least basic first, most basic last) C...
A:
Q: In colligative properties, the unit of concentration that is employed is which is number of moles of...
A: Colligative properties are physical properties of solution.
Q: Gold occurs in the ocean in a range of concentrations of 0.1 to 2 mg gold per ton of seawater. Near ...
A: Given recovery = 64% 1 troy = 31.1g Step 2 Recovery us 64% So amount of gold that can be rec...
Q: Which is more reactive for electrophilic addition ?
A: The compounds given are,
Q: Describe the following properties of chromatogram (including formula if applicable): Bandwidth Rete...
A: 1.Bandwidth sets the frequency around the wavelength provided by the detector to view and report chr...
Q: Consider the following sets of quantum numbers (n, , mj, ms). Determine which are incorrect and stat...
A:
Q: According to the following reaction, how many grams of nitrogen gas will be formed upon the complete...
A:
Q: A solution has [H+]=4.0x10-8M. Determine the POH of the solution
A:
Q: Can acetic acid be used as the mobile phase in kool aid chromatography over isopropyl alcohol? Desc...
A: Kool aid chromatography involves separation of red and blue dyes present in the Kool aid. The mobile...
Q: Which of the following statements is TRUE? Select one: a. Molecule on the left is E, molecule on the...
A: Option (b) is correct. Assignment of E/Z configuration is generally used for the alkenes. E stands...
Q: For this experiment, yield was not important, but in onsite lab, as well as large-scale processes, a...
A: The solution is given below -
Q: 10 Temperature (°C) CHAPTER 5 MULTIPLE-CHOICE QUESTIONS Questions 1-5 use the following information....
A: We are given the heating curve of a substance as follows:
Q: What error would occur if the Sep-Pak C18 cartridge (the nonpolar stationary phase) was not subjecte...
A: C-18 cartridges are columns that are filled with packing material that act as stationary phase. The ...
Q: What is the crystalline structure of Chromium? HCP Cubic BCC FCC
A: Crystalline structure of Chromium:
Q: Identify one chemical reaction that changed history and explain how it influenced modern day industr...
A: A question based on general chemistry that is to be accomplished.
Q: 20-g of benzoic acid is dissolved in 600 mL of water. Calculate the pH of the solution.
A: Since you have posted multiple questions, we are entitled to answer the first only. 1) Given: Mass o...
Q: A Moving to another question will save this response. Question 4 A sodium chloride solution is 38% N...
A:
Q: A lemon-flavored drink contains an unknown amount of citric acid, C5H8O7. What is the molar concentr...
A: Given :- C5H8O7 + 3NaOH --> Na3C5H5O7 + 3H2O Volume of C5H8O7 solution = 100.0 mL Molarity of ...
Q: [Co(H2O)6l2* 19400 16000 8000 25 000 20 000 15 000 10 000 5 000 Frequency (cm1) Answer the following...
A: (i) The ground states and excited states of the same multiplicity for free metal ion is given below-...
Q: Assuming that solutions of the given compounds are equal in concentration, which of the following is...
A: In identical conditions, more number of ions after dissociation means conductivity should be more.
Q: A Click Submit to complete this assessment. Question 20 6 p Using orbital diagrams, determine the nu...
A: Electronic configuration: It is the distribution of electron in the atomic orbitals of the element.
Q: What is the concentration of the bromide ion if 25.0 ml of a 0.50 M AlBr3 solution combines with 40 ...
A: Given, Concentration of AlBr3 solution = 0.50 M = 0.50 mol/L Volume of the AlBr3 solution = 25.0 mL ...
Q: The degree of dissociation of Ca(NO,)2 in a dilute aqueous solution containing 14 g of salt per 200 ...
A:
Q: Describe how you would prepare a) 2.50L of 0.225M sodium hydroxide solution starting with the commer...
A: Concentration of a solution is the amount of solute present in fixed amount of solvent or solution. ...
Q: 1) H3O* 2) concentrated H2SO4, heat 3) Оз 4) DMS
A:
Q: For the following conceptual problems, provide written responses with your selection of the molecule...
A:
Q: Which of the following statements are correct? Both of these. Rearrangement is a property of carbani...
A: Carboanion is the species in which carbon carries negative charge. Carbonium ion is the species in w...
Q: Fonacha of 5.50mol/L.whet. A stock solution has a "concentrdion volune ef this stock Solution is req...
A:
Q: onsider the reaction 2N2O5(g) → 4NO2(g) + O2(g). Use the time-monitored concentration data to verif...
A:
Q: Find the stereocenters in penicillin: R. -S CH3 -N- CH3 СООН IZ
A:
Q: Determine the oxidation number of the Chromium (Cr) in an unknown salt if electrolysis of a molten s...
A:
Q: A bag of popcorn that had a mass of 110.0g. After popping the bag its mass is 100.0g. After consumin...
A: Percentage of water can be calculated by dividing the mass of water to mass of popcorn into 100.
Q: For the redox reaction given below complete the following: Mn²- - NaBiO, – Bi?- + MnO4 (a) What is t...
A: Ans A . Oxidation state of Bi in NaBiO3 = 5 Ans B . Oxidation state of Mn in MnO4- = 7 Ans C . Mn is...
Give the mechanism and product of the following reaction
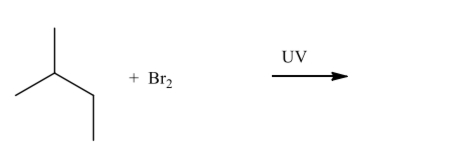
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The concentration of methylene blue in the sample is estimated through (select the correct) options: refractive index measurement (refractometry). turbidity measurement (turbidimetry). absorbance measurement (spectrophotometry). titration with 2% HCl solution.Kindly answer question i, ii & iiichemhelp7 The researcher determined the Pb content of water sample from a river suspected to be contaminated with Pb from a nearby factory by complexing with cyanidine and the absorbance measured was 0.405. What is the lead content of the water sample? a. 4.23 ppm b. 4.28 ppm c. 207 ppm d. 333 ppm

