We need to find an element that is able to have a lot of un-paired electrons. The more degenerate (same energy) orbitals a sublevel/subshell has, the more unpaired electrons the atom can contain. The periodic table below has highlighted all of the elements that are filling electrons into sublevels/subshells with degenerate orbitals (multiple orbitals of equal energy, such as the three p orbitals in a p subshell). PERIODIC TABLE OF THE ELEMENTS H 14 16 VIA Не 17 NA VIA 4.00 Li Ве B CNOF Ne Na Mg 2Al Si P S CIA. 22 2430 19 20 K Ca Sc Ti v Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 30.0 37 38 Rb Sr Y Zr Nb Mo Te Ru Rh Pd Ag Cd In Sn Sb Te I Xe- 56 Cs Ba "La Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn IN 87 Fr Ra 'Ac Rf Db Sg Bh Hs Mt Ds Rg (22 202as *Lanthanide Series Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 4012 4 2 4 7 9 "Actinide Series Th Pa Ux Pu Am Cm Bk Cf Es Fm Md No Lr 100 101 102 10 Explain which regions of the periodic table would be best for holding multiple unpaired electrons. Make sure to discuss what SU subshell is being filled with electrons in each of those regions of the periodic table
We need to find an element that is able to have a lot of un-paired electrons. The more degenerate (same energy) orbitals a sublevel/subshell has, the more unpaired electrons the atom can contain. The periodic table below has highlighted all of the elements that are filling electrons into sublevels/subshells with degenerate orbitals (multiple orbitals of equal energy, such as the three p orbitals in a p subshell). PERIODIC TABLE OF THE ELEMENTS H 14 16 VIA Не 17 NA VIA 4.00 Li Ве B CNOF Ne Na Mg 2Al Si P S CIA. 22 2430 19 20 K Ca Sc Ti v Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 30.0 37 38 Rb Sr Y Zr Nb Mo Te Ru Rh Pd Ag Cd In Sn Sb Te I Xe- 56 Cs Ba "La Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn IN 87 Fr Ra 'Ac Rf Db Sg Bh Hs Mt Ds Rg (22 202as *Lanthanide Series Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 4012 4 2 4 7 9 "Actinide Series Th Pa Ux Pu Am Cm Bk Cf Es Fm Md No Lr 100 101 102 10 Explain which regions of the periodic table would be best for holding multiple unpaired electrons. Make sure to discuss what SU subshell is being filled with electrons in each of those regions of the periodic table
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter11: Modern Atomic Theory
Section: Chapter Questions
Problem 126CP
Related questions
Question
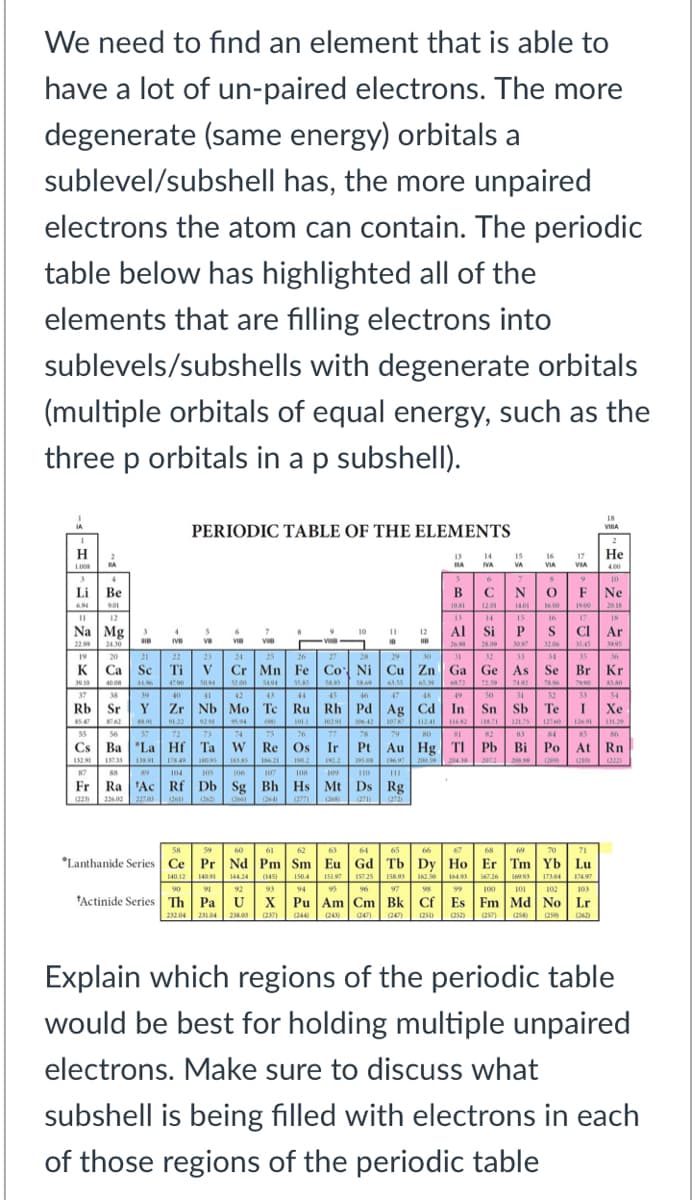
Transcribed Image Text:We need to find an element that is able to
have a lot of un-paired electrons. The more
degenerate (same energy) orbitals a
sublevel/subshell has, the more unpaired
electrons the atom can contain. The periodic
table below has highlighted all of the
elements that are filling electrons into
sublevels/subshells with degenerate orbitals
(multiple orbitals of equal energy, such as the
three p orbitals in a p subshell).
18
PERIODIC TABLE OF THE ELEMENTS
H
Не
NA
VA
VIA
CNO
140 .00 0 20.18
4 IS
Li Be
B
F Ne
Na Mg
22.99
12AI Si PSCIAr
10
19
K Ca Sc Ti v Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr
53 54
Rb Sr Y Zr Nb Mo Te Ru Rh Pd Ag Cd In Sn Sb Te1 Xe
Cs Ba La Hf Ta w Re Os Ir Pt Au Hg TI Pb Bi Po At Rn
Fr Ra 'Ac Rf Db Sg Bh Hs Mt Ds Rg
*Lanthanide Series Ce Pr Nd Pm Sm Eu Gd Tb| Dy Ho Er Tm Yb Lu
14424 (145)
92
tActinide Series Th Pa U
15197 15725 158.93 162.50 16493
95 96 97 9 100 101 102 103
Pu Am Cm Bk| Cf Es Fm Md No Lr
140.12 140
150.4
17497
93
94
X
232.04 2104 2.05
(237)
(244
Explain which regions of the periodic table
would be best for holding multiple unpaired
electrons. Make sure to discuss what
subshell is being filled with electrons in each
of those regions of the periodic table
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning