What is the charge of each amino acid residue in the peptide chain and what is the net charge on the whole protein at pH = 4 (Use the provided table)? KGL
What is the charge of each amino acid residue in the peptide chain and what is the net charge on the whole protein at pH = 4 (Use the provided table)? KGL
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter23: Organic Polymers, Natural And Synthetic
Section: Chapter Questions
Problem 44QAP: A 1.00-mg sample of a pure protein yielded on hydrolysis 0.0165 mg of leucine and 0.0248 mg of...
Related questions
Question
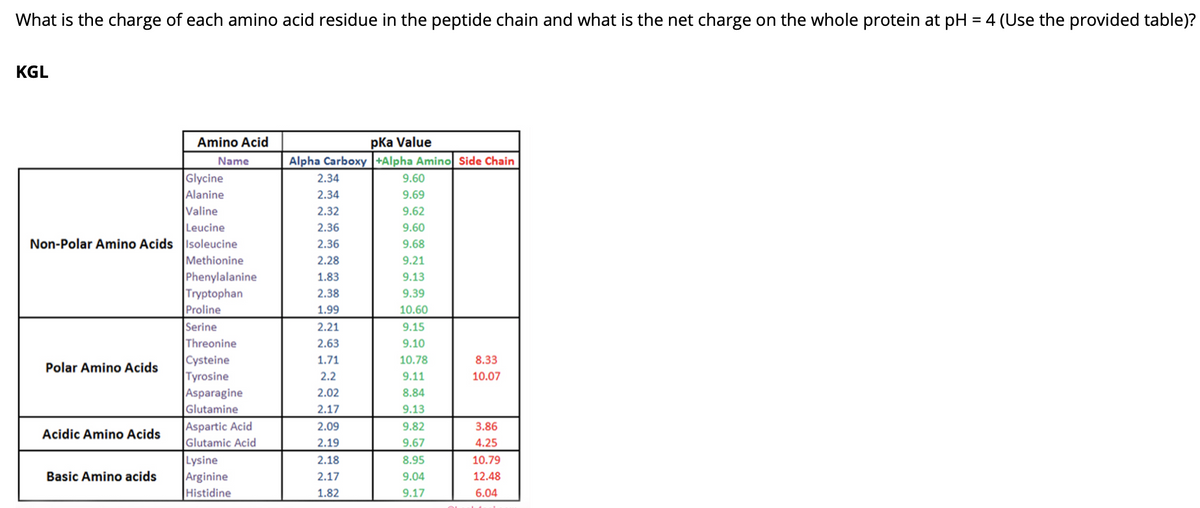
Transcribed Image Text:What is the charge of each amino acid residue in the peptide chain and what is the net charge on the whole protein at pH = 4 (Use the provided table)?
KGL
pka Value
Alpha Carboxy |+Alpha Amino Side Chain
Amino Acid
Name
Glycine
Alanine
Valine
2.34
9.60
2.34
9.69
2.32
9.62
Leucine
2.36
9.60
Non-Polar Amino Acids Isoleucine
Methionine
Phenylalanine
Tryptophan
2.36
9.68
2.28
9.21
1.83
9.13
2.38
9.39
Proline
1.99
10.60
Serine
Threonine
Cysteine
Tyrosine
Asparagine
Glutamine
2.21
9.15
2.63
9.10
1.71
10.78
8.33
Polar Amino Acids
2.2
9.11
10.07
2.02
8.84
2.17
9.13
Aspartic Acid
Glutamic Acid
Lysine
Arginine
2.09
9.82
3.86
Acidic Amino Acids
2.19
9.67
4.25
2.18
8.95
10.79
Basic Amino acids
2.17
9.04
12.48
Histidine
1.82
9.17
6.04
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning
