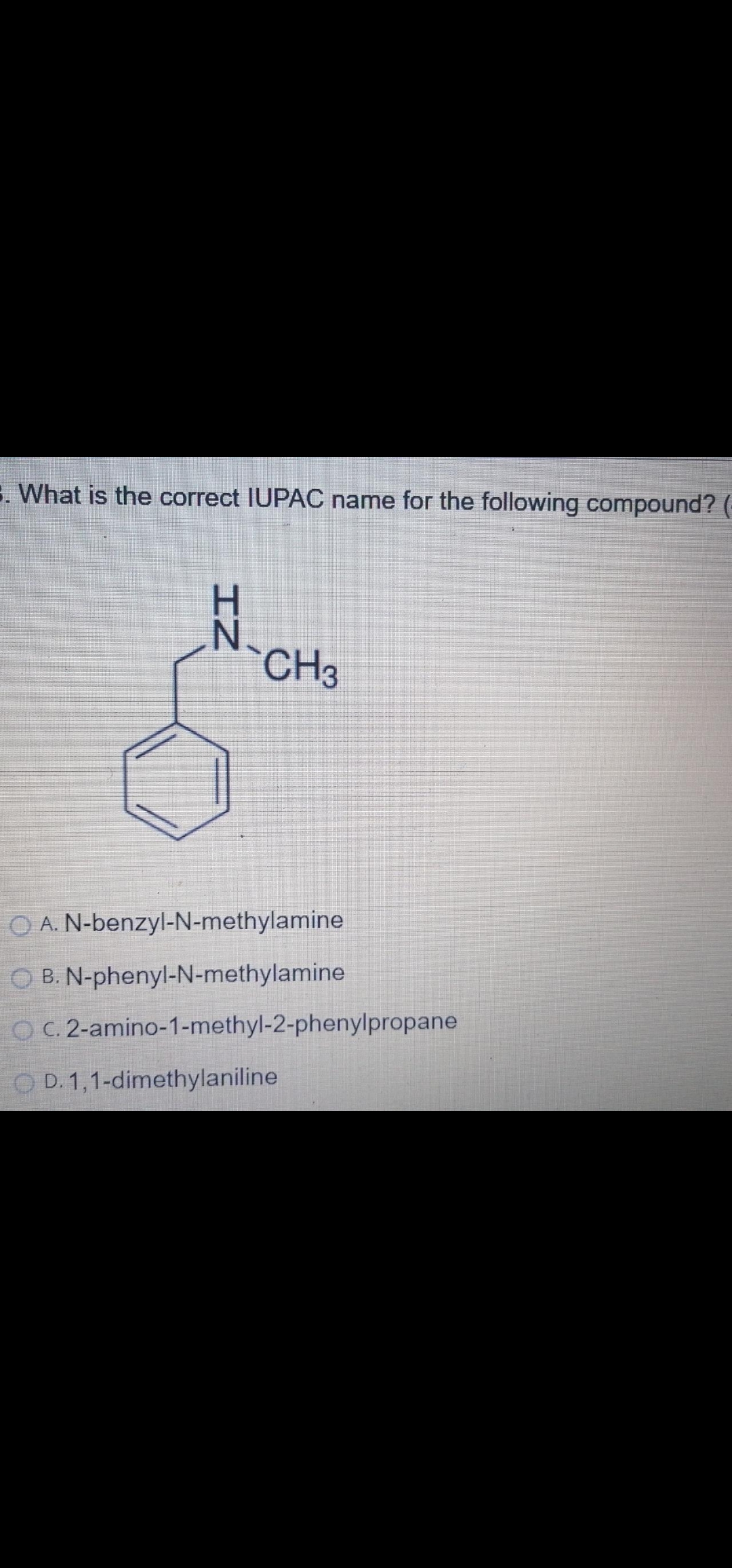
What is the correct IUPAC name for the following compound? H. N. CH3 O A. N-benzyl-N-methylamine O B. N-phenyl-N-methylamine O C. 2-amino-1-methyl-2-phenylpropane O D. 1,1-dimethylaniline
Q: General Formula (Low molecular Weight) Туре of Compound Polarity solubility in Choices Solubility in…
A: Solutions -
Q: 10. Answer the following briefly: A. What class is the hydroxyl group (-OH) used in? B. What does -X…
A: Organic compounds can be defined as substances in which carbon atoms are either attached to…
Q: Match column A (common active ingredients of insect repellants) to column B (IUPAC names). Column A…
A:
Q: 4 A certain carboxylic acid belonging to the homologous series of aliphatic saturated monocarboxylic…
A: General formula of a aliphatic saturated carboxylic acid is R-COOH, where R is alkyl group. Given…
Q: For Fehling's Test not all aldehydes reacts to this test, what are the limitations and does the…
A: Fehling solution or reagent is used to distinguish between aldehyde and ketone other than alpha…
Q: Chemistry Draw these components semi-developed a) pentan-2,3,4-trione b)heptyl ethanoate c)…
A:
Q: Benzylpenicillin shown in Figure 34 was the first antibiotic to be manufactured on large scale. How…
A: The solution is as follows:
Q: What type of test/s should we use to differentiate aliphatic aldehydes from aromatic aldehydes?…
A: Aliphatic aldehyde and aromatic aldehyde can be distinguished.
Q: Is it possible for Primary and Secondary Alcohols to undergo Nucleophilic Addition? How about…
A: Given: Primary and Secondary Alcohols reaction :
Q: Draw the structure of another product that could form in the reaction mixture?
A: When benzophenone reacts with 2- propanol in presence of light and alcohol it will form benzopinacol…
Q: Test used to determine secondary alcohols with a methyl group attached to the carbon where the…
A: Iodoform test is used to detect the presence of carbonyl compounds with the structure R-CO-CH3 or…
Q: Mark the reactions in Figure 21 that will produce an alcohol as the main organic product. *…
A: Alcohol can be produced in many ways Alcohol are organic compound which have OH moiety as functional…
Q: The following name is incorrect according to IUPAC systematic rules. 2,2-Diethylbutane State which…
A: In this name the alphabetical listing by groups rule is correct . The punctuation rule is also…
Q: +||+ 39. This aldehyde is useful for its vanilla flavoring hence the trivial name, vanillin. A. B.…
A: ✓Vanillin is an organic compound with the molecular formula C8H8O3. It is a phenolic aldehyde. Its…
Q: te
A: For the nomenclature of organic compounds we follow certain rules ; 1) choose a parent chain (the…
Q: A B он NO2 но D
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: Give an IUPAC name for the substances below: ball & stick- 2nd structure: labels ball & stick labels…
A: IUPAC nomenclature follows following rules in these compounds:-
Q: CH3- CH2- CH2- Br KMNO, sOCIz A B (c AIBR, H20
A:
Q: Why wash with sodium hydroxide? A) It's known to break down and solubilize hydrocarbons (oven…
A: Sodium hydroxide is a water-soluble strong base.
Q: Oraw Figure 2 in the space below, the synthesis of melatonin, for each step label the functional…
A: Given that, the synthesis of melatonin from tryptophan. We have to level the functional groups for…
Q: WRITE THE IUPAC NAME OF THE FOLLOWING:
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: Question 38: Consider the synthesis scheme in Figure 38. What type of compound is represented by C…
A: Please find your solution below : The reaction of benzene with propyl bromide in presence of AlBr3…
Q: A. What is scum? Why would it form in in hard water but not soft water? B. (Synthetic) detergents…
A: Scum : Hard water is having calcium, Mg salts. These salts react with soap and forms insoluble…
Q: Is chlorophyll soluable in 2-propanol? Why or why not?
A: Chlorophyll is a large carbon chain molecule with an ionic tail and molecular formula C₅₅H₇₂O₅N₄Mg…
Q: What is the functional group of propanoic acid? How do we know if it's soluble in water or insluble…
A: Qualitative analysis is a technique to identify the species present in the given unknown sample. The…
Q: where's the usual country of origin of hydrochloric acid 37%? where's the usual country of origin of…
A: 1) Hydrochloric acid(HCl) - is also known as muriatic acid. It is colorless solution . Used as…
Q: Fill-up in the blanks: Common Name 1. N-tert. pentyl- valeramide 2. (5) IUPAC Name (1)…
A: IUPAC nomenclature -The International Union of Pure and Applied Chemistry (IUPAC) nomenclature of…
Q: Which of water or hexane is the better solvent for: A) C10H22 B) CH3CH2NH2 C) K3PO4
A: Given Compounds, (A) C10H22 (B) CH3CH2NH2 (C) K3PO4
Q: Oxidation of Butanol -1 in KMnO4/H+ will produce-------------.
A: Potassium permanganate is one of the most important oxidizing agents which is widely used in the…
Q: H + H₂O H₂SO4 →
A:
Q: Part E OH HO Enter the correct IUPAC nomenclature.
A: The IUPAC nomenclature of the given compound has to be provided. The given compound is,
Q: Which should be least soluble in water? Н H-C-0-H methanol H H II H-N-C-H methylamine H H H 01H Η T…
A:
Q: why CH3CONH2 is a stronger acid and a weaker base than CH3CH2NH2?
A:
Q: Give a clear handwritten answer of this reaction...Give the IUPAC name all reactions given bleow...?
A: To find : The IUPAC name all reactions .
Q: To oxidize an alkyl benzene structure we need at least one benzyllic hydrogen. So think this…
A: The details solution for this is provided below in attach image.
Q: For each pair, give the relationship between the two compounds. Making models will be helpful.(a)…
A:
Q: Question 7. why methylbenzoate a liquid whereas benzoic acid (having lower molecular mass) is a…
A: Different intermolecular forces are found between molecules that depends on the type of molecule and…
Q: one greener approach for synthesising succini
A:
Q: (b) Phenol, benzene, chlorobenzene, benzoic acid (c) Benzene, bromobenzene, benzaldehyde, aniline
A:
Q: Answer the following questions based on the reaction below: a) State reagent D. b) Draw the…
A: The details solution for this is provided below in attach image.
Q: 3. What is one chemical test that can distinguish between 3-hexanol and 3-hexanone? Indicate the…
A: Chemical test to distinguish between 3-hexanol and 3-hexanone is reaction with…
Q: QUESTION 30 Naming symmetrical anhydrides is easy! 9 In class, we learned to find the parent acid,…
A: Anhydride have the general structure - R1 ----CO----O----CO------R2 If R1= R2 than the anhydride…
Q: Test used to determine secondary alcohols with a methyl group attached to the carbon where the…
A: Iodoform test is used to detect the presence of carbonyl compounds with the structure R-CO-CH3 or…
Q: Write down a chemical name of Benzaldehyde?
A: Given compound is : Benzaldehyde Chemical name of Benzaldehyde = ?
Q: TRUE OR FALSE; an sp2 carbon may exist in an aldehyde.
A:
Q: Complete the table below with the following choices. Answer those with numbers 81 to 90. A.…
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of…
Q: Fractional distillation lab experiment for organic chemistry What are possible other identification…
A: Distillation is a powerful tool to identification and purification of organic compounds.
Q: Give examples of chemical reactions for both saytzeef product and hofmann product. state the iupac…
A: When a removing group or Molecule removed from the compound, alkyl halide then , the removal take…
Q: The alcohol below is a ________________ alcohol and there are _____________ possible different…
A: Primary alcohol = If OH group is attached to that carbon ,which is further attached to 1 carbon .…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- based on this video: on Hinsberg Test Tests for Amines - MeitY OLabs (8.5min) https://www.youtube.com/watch?v=j5jgMUWri8U Write the reaction (two-step, in skeletal) of each test amine when tested in the Hinsberg Test.COMPLETE THE TWO REACTIONS BELOWIs the crude (non-crystallized) acetanilide pure? Why or why not? Is the crystallized acetanilide pure? Why or why not?
- as a follow-up question can explain this mechanismFrom benzene, organic compunds with 5 carbonw or less and any other inorganic compound you want. Synthesize the derivative for the naphthalene.Chemistry Please answer the following two questions. 1.) Why are functional group interconversions controversial? (double bonds into epoxide) 2.) Explain the clincal trials that happen for epoxide containing molecules
- When aspirin is made from acetic acid, the by product is H2O; when acetic anhydride 1Sused, the by-product is acetic acid. When acetyl chloride is used (see above), the by product is HC1. Which route should you use if you were working for Leica, a company that mass produces aspirin on a daily basis? Why?What is the Rf of your alcohol product B? Which fractions would you combine to isolate pure alcohol product B?Consider a mixture of the following molecules. Which molecule will be in the organic layer after treatment with excess acid (e.g. HCl)? Options: A B C D All of the above
- Chemistry Please help explain this textbook question: Although N, N -dimethylaniline is extremely reactive toward electrophilic aromatic substitution and is readily substituted by weak electrophiles, such as diazonium and nitronium ions, this reactivity is greatly diminished by the introduction of an alkyl substituent in an ortho position.What is the correct reaction scheme look like between acetic acid and 3-methylbutanol in the presence of sulfuric acid? Which image attached is the correct reaction scheme?Given this organic synthesis, is there any limiting and excess reagents? also describe the procedure is run, how is the reaction monitored?Is the order fo addition important? N-ETHYLALLENIMINE[Aziridine, 1-ethyl-2-methylene-] Submitted by Albert T. Bottini and Robert E. Olsen1.Checked by Thomas H. Lowry and E. J. Corey. 1. ProcedureCaution! This preparation should be carried out in a good hood to avoid exposure to ammonia. The operator should wear rubber gloves and protective goggles because 2-haloallylamines and ethylenimines can cause severe skin and eye irritation. A 2-l. three-necked flask is fitted with a sealed mechanical stirrer, a gas-inlet tube, and a dry ice condenser protected from the air by a soda-lime drying tube (Note 1). The system is flushed thoroughly with dry ammonia, and 32.8 g. (0.84 mole) of sodium amide (Note 2) is added to the flask. The system is again flushed with ammonia, the condenser is provided with dry ice covered by acetone, and 1.2 l. of liquid…

