Q: 5. Draw a structural example of a primary, secondary and tertiary alcohol.
A:
Q: 9. For the reaction: 2 CH3OH(g) + H2(g) CaH«(g) + 2 H;O(g) a) Calculate AG° for this reaction using…
A:
Q: D. Enthalpy is negligible B. Enthalpy change is negative 7. Which represents an exothermic reaction?…
A: ∆H = ∆H(product) - ∆H(reactant) -> In exothermic reaction heat is released that means ∆H = -ve…
Q: A chemist prepares a solution of sodium chloride (NaCl) by measuring out 33.6 g of sodium chloride…
A: given :- mass of NaCl = 33.6 g volume of solution = 100. mL to calculate :- concentration of…
Q: counts per minute
A:
Q: An analytical chemist is titrating 206.5 mL of a 0.9600M solution of cyanic acid (HCNO) with a…
A: Given- Volume of HCNO= 206.5 mL Concentration of HCNO= 0.9600M Concentration of KOH=1.100M Volume…
Q: Write the empirical formula for at least four ionic compounds that could be formed from the…
A:
Q: e formula of ionic compounds
A:
Q: gummy bear has a mass of about 2.3g and a volume of about 1ml. About 12% of the mass of the gummy…
A: Given-> Weight of gummy bear = 2.3 gm Volume = 1 ml = 0.001 L (1L = 1000 ml) Weight of sugar =…
Q: Which of the following molecules would be the least polarizable? O CH3CH2CH3 O SIH3SIH3 O CH3CH3 O…
A: Polar molecule: Polar molecules will form when two atoms don't share electrons equally in a covalent…
Q: 1o A 5 cm sample of 0.05 mol dm3 sodium chloride is mixed with a 5 cm3 sample of 0.05moldm potassium…
A:
Q: A Zn wire and Ag/AgCl reference electrode saturated with KCI (E = 0.197 V) are placed into a…
A: Given data :
Q: Which of the following is a secondary amine? NH2 NH -NH2 d) `NH2 а) b)
A:
Q: What is the sign for each of the following? (Change in) a. S, H, G for the evaporation of a…
A:
Q: -tBuo O-Bu H,N. HO, + PBUOH + CO, NEt, CH,CI, Ph
A: Note : Boc protection of amines.
Q: At 122°C the pressure of a sample of nitrogen 1.07 atm. At 205°C the pressure will be ____ atm ,…
A: We need to find the final pressure or the final temperature according to the given data
Q: Draw the major organic product of the reaction conditions. Show stereochemistry in your structure.…
A: Major organic product of the given reaction = ?
Q: Which is true for the endothermic reaction 2 SO3(g) → 2 SO,(g) + 02(g)? A) Spontaneous at all T B)…
A: According to the second law of thermodynamics, the spontaneous nature of the reaction is determined…
Q: A 500.0-mL sample of H2 gas is collected OVER WATER at 298 K and 747 torr. What volume would the…
A:
Q: Describe the relationship between atoms, elements, and matter
A:
Q: Using molarity to find solute mass Iculate the volume in liters of a 0.208 mol/L copper(II) sulfate…
A: Given -> Molarity = 0.208 mole/L Weight of CuSO4 = 75.0 gm
Q: 2. Conversion. Using the moving the decllalI a. 60 mg b. 0.005 L mL c. 200 ml kg
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for…
Q: Which of the following metal is used for the hydrogenation of benzene? a Iron b Copper c…
A: Hydrogenation is an addition reaction in which hydrogen atoms are to the benzene ring and forms…
Q: What is the H3O* | and the pH of a benzoic acid-benzoate buffer that consists of 0.13 M C6H5COOH and…
A: Given: Concentration of C6H5COOH = 0.13 M Concentration of C6H5COONa = 0.30 M Ka of C6H5COOH =…
Q: A buffer is prepared by mixing 206 mL of 0.452 M HCl and 0.500 L of 0.400 M sodium acetate,…
A: Given: Molarity of HCl = 0.452 M Volume of HCl = 206 mL Molarity of sodium acetate = 0.400 M Volume…
Q: What would be the product of this reaction? A • A- •B - •C - D- •E-
A:
Q: What is the voltage for a cell that has a Zn electrode in a 1.0 M aqueous Zn2+ solution on the left…
A: Voltage of cell can be calculated using nernst equation.
Q: What is the IHD of a chemical compound with a formula of C6H5NO2? a 4 b 5 c 6 d 7
A: IHD stands for Index of Hydrogen Deficiency. In a hydrocarbon with no double bond or rings, the no…
Q: Check any and all boxes to indicate which reagents can be used to carry out this transformation…
A: the reaction is reductive ozonolysis, where alkene is converted to aldehyde and ketone group,in…
Q: 3. Suggest experimental means by which the rates of the following reactions could be followed. There…
A: In the question it is given to discuss the experimental means by which the rates of the reaction…
Q: Ksp for Fe(OH)3 (Fe(OH)3(s) ⇌ Fe3+(aq) + 3OH-(aq)) is 4.0 × 10-38. In the presence of 0.01 M NaOH,…
A:
Q: Predict the product of each reaction below and indicate if the mechanism is likely to be SN1, Sx2,…
A:
Q: the voltage of the cell is 0.09223 V at 25.0°C when [Cd 2+( aq)] = 0.1036 M and the pressure of H 2…
A: Given- Voltage of the cell= 0.09223 volt [Cd2+(aq)]=0.1036M E°=0.4030V Pressure of H2=1atm pH=?…
Q: Part A Electrolytic cells use electricity to cause a nonspontaneous redox reaction to occur. An…
A: We have find out the answer.
Q: Calculate the pH of the following solutions: 0.0035 M HBrO solution (K,=2.5x10) 0.0624 M H,CO,…
A:
Q: 1. PBr, 2. NaNg, THF HO 3. Pd/C, H2 (Excess) 4. Mel (Excess) 5. Ag,0, H20 `NH2 NH2 A B D
A: For the given sequences of reaction of the given alcohol, we can predict alkene as the major…
Q: For the following reaction, 12.7 grams of chlorine gas are allowed to react with 58.1 grams of…
A:
Q: 4-What is the difference between laboratory and point-of-care or at-home cholesterol tests?
A: The difference of cholesterol test in laboratory and point of care or at home will discuss according…
Q: The heat of vaporization AH, of methanol (CH,OH) is 35.3 kJ/mol. Calculate the change in entropy AS…
A:
Q: Problem: CO2 gas occupies a volume of 20.0 L at STP. How many moles and How many molecules are…
A: According to the ideal gas equation, the volume of a gas depends on the temperature, pressure, and…
Q: How many electrons can the third principal quantum level hold? [x] Explain how you got the answer in…
A: There are 4 different types of quantum numbers- 1- principal quantum no 2- azimuthal quantum no…
Q: For the following reaction, 6.87 grams of iodine are mixed with excess hydrogen gas . The reaction…
A:
Q: Be sure to answer all parts. Determine the percent ionization of the following solutions of phenol…
A:
Q: Given is teh mass spectogram of an unknown compoound. This unstable cyclic compund is a colorless…
A: The compound name is cyclohexene. Cyclohexene goes retro aldol produces the m/z = 54.
Q: What is the component concentration ratio, [Pr−]/[HPr], of a buffer that has a pH of 4.98? (Ka of…
A:
Q: What is the major driving force in chromatographic separation? difference in affinity with a…
A: Chromatography has been defined primarily a separation process which is used for the separation of…
Q: Draw the product of the following reaction. CH3 H2N HOʻ ČH3 но он NaCNBH, OH Он
A: Sugar is having a aldehyde group. Therefore it reacts with amine forms imine. Imine is converted…
Q: Be sure to answer all parts. Calculate the solubility in g/L of AgBr in (a) pure water and (b)…
A:
Q: o bonds: a bonds:
A:
Q: 8 The illustration to the left represents a mixture of phosphorus ( orange ) and fluorine ( green )…
A: Answer: Limiting reagent is that reactant which exhaust first in chemical reaction and decides the…
Step by step
Solved in 2 steps

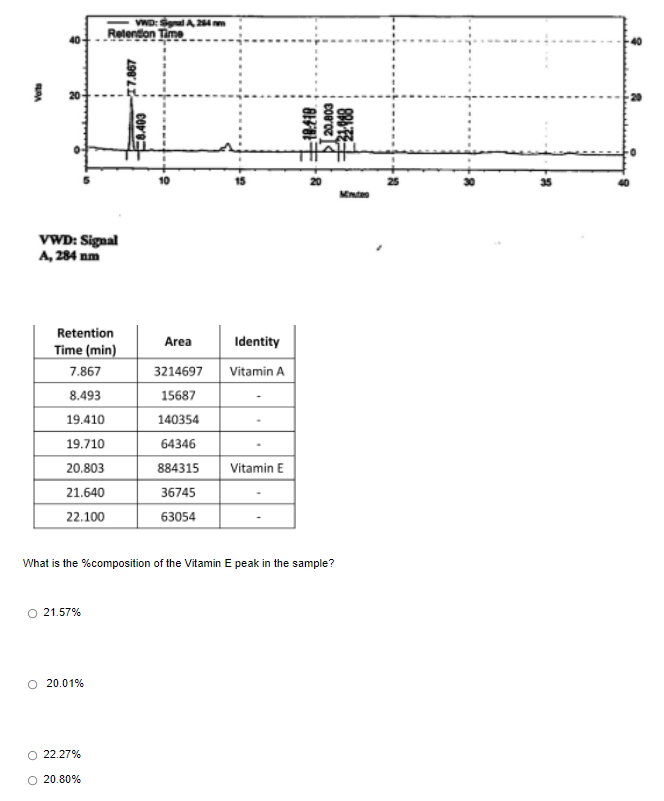
- In order to improve the peptide separation by using a HPLC system, trifluoroacetic acid acts as mobile-phase modifier was added during the preparation of mobile phase. The preparation was performed by a postgraduate student as following:“2.851 g trifluoroacetic acid (MW: 114.02 g/mol) was made up to 500 cm3 in a graduated flask. To this solution, 50 cm3 of ethanol was added, and after mixing the mobile phase was placed in the solvent reservoir and pumping was commenced at 1.5 cm3 min-1.”Based on the given preparation procedure, identify THREE mistakes that were made.A 0.0200 gram blood sample was decomposed by a microwave digestion technique followed by dilution to 100.0 mL in a volumetric flask. Aliquots of the sample solution were treated with a lead complexing reagent and water as follows: Solution 1: 10.0 ml blood sample + 20.0 mL complexing agent + 30.0 mL H20. Solution 2: 10.0 ml blood sample + 20.0 mL complexing agent + 26.0 mL H20 + 4.00 mL of 78 ppb Pb2+ standard. The resulting solutions were analyzed by UV/Vis at 375 nm. Absorbance for solution 1 = 0.155 and for solution 2 = 0.216. Calculate the concentration of lead (ppb) in the original sample.analyte concentration(C)(mg/ml) injection volume (ul) elution time (time) peak DAD signal(mAU) caffeine 1 1 4.67 302.85 aspartame 5 1 7.53 15.83 benzoic acid 1 1 8.14 89.98 saccharin 1 1 1.91 84.86 mixture(add everything above with 1:1:1:1 ratio) 1 4.47 69.58 How to get the concentration of the mixture in this case?
- The following is an HPLC separation of deuterated benzenes on a C18 column that 440 cm long. The mobile phase was 30% acetonitrile:70% water at 30oC. What is the mobile phase velocity? Find the retention factor k' for C6D6. Find the plate number and plate height for C6D6.. Assuming that the peak widths for C6H5D and C6H6 are the same as that of C6D6., find the resolution for C6H5D and C6H6. Retention times for C6H5D and C6H6 are 193.3 and 194.3 respectively, Find the relative retention time and unadjusted relative retention time between C6H5D and C6H6.Column Plate Number: 16Retention Factor: 3.1Sample Peak Retention Time: 1.2minutesSample Peak Width: 1.53First Peak Retention Factor: 2.10Second Peak Retention Factor: 1.37Efficiency: 0.43Pressure: 375barrColumn: 28cm x 5mmViscosity: 2.3poiseFlow Rate: 1.2Column Permeability: 2.8 FIND: a & ba. Resolution b. Pressure if flow rate is adjusted to 2.3 c. Particle Size d. Selectivity Factor e. The retention time of the unretained peakYou obtained the following raw data when setting up a Bradford standard curve: BSA (mg/ml) Absorbancy 595nm 0 0.225 1 0.310 2 0.420 3 0.510 4 0.610 5 0.720 6 0.810 7 0.915 8 0.950 9 0.980 10 0.990 After blanking against a bradford-dH2O sample, the protein concentration of an unknown sample was determined using the same method and an absorbancy of 0.523 was obtained. Set up a standard curve, excluding outliers (experimental and statistical) and determine the protein concentration in the unknown sample in mg / ml (up to 3 significant figures).
- PART A: Substance A was found to have a retention times of 3.15 minutes on a 70.00-cm column. An unretained species passed through the column in 0.50 minutes. The peak width (at base) for A was 0.60 minutes. Using this information, determine the following: 1. What is the retention factor for analyte A? 2. What is the plate count for this column? 3. What is the height equivalent of a theoretical plate? PART B: Match each statement with one of these terms. A Partition coefficient B Mobile phase C Elution D Longitudinal diffusion E Stationary phase F Eddy diffusion G Retention factor H Plate number I Dead time J Retention time K Selectivity factor L Column resolutionWhat is the retention factor of a sample when after injectiion the void time (tM) took 4.18 sec? The retention time (TR) is 21.19 secFT-IR technique can be utilized for the analysis of unknown analytes by matching it with__________. Both choices are correct Commercial library/database Reference standard
- Let the retention factor of species A be 0.5, while the retention factor of species B be 0.3. If species A moves 1.0 cm how far does species B move? 0.60 cm 0.80 cm 1.0 cm 1.2 cm 1.6 cmTwo species A and B are known to have water/hexane partition coefficient of 5.99 and 6.16.They are separated by elution on silica gel with hexane as eluent. The ratio for the packingVS/VM =0.425a. Calculate the retention factor for each soluteb. Calculate the selectivity factorc. How many plates are needed to provide a resolution of 1.5?d. What column length should be used to provide a resolution of 1.5 if the plate height ofthe packing is 1.5 ×10-3cm?e. If the flow rate is 6.75 cm min-1, how long will it take to elute the two species? (the question wanted to be answered)A 5.00-mL sample of blood was treated with trichloroacetic acid to precipitate proteins. After centrifugation, the resulting solution was brought to pH 3 and extracted with two 5-mL portions of methyl isobutyl ketone containing the lead-complexing agent APCD. The extract was aspirated directly into an air/acetylene flame and yielded an absorbance of 0.527 at 283.3 nm. Five-milliliter aliquots of standard solutions containing 0.400 and 0.600 ppm of lead were treated in the same way and yielded absorbances of 0.396 and 0.599. Find the concentration of lead in the sample in ppm assuming that Beer’s law is followed.





