Absorbance at 210 nm →→ Thiourea (unretained) t = 41.7 min H₂N S NH2 188.1 D min D D D 4 5 D W1/2= 1.01 min 2 Ал At= 1.01 min 185 190 195 Time (min) 5 4 100 200 Time (min) D 300 CD3 CD3 CH3 7 8 D
Absorbance at 210 nm →→ Thiourea (unretained) t = 41.7 min H₂N S NH2 188.1 D min D D D 4 5 D W1/2= 1.01 min 2 Ал At= 1.01 min 185 190 195 Time (min) 5 4 100 200 Time (min) D 300 CD3 CD3 CH3 7 8 D
Chapter13: Structure Determination: Nuclear Magnetic Resonance Spectroscopy
Section13.SE: Something Extra
Problem 31AP: When measured on a spectrometer operating at 200 MHz, chloroform (CHCl3) shows a single sharp...
Related questions
Question
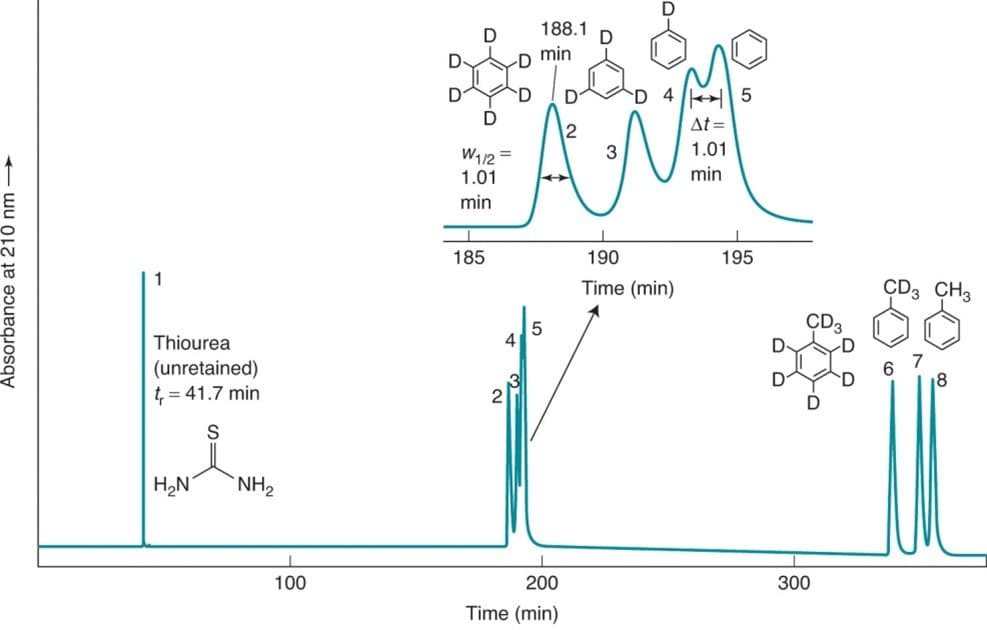
- The following is an HPLC separation of deuterated benzenes on a C18 column that 440 cm long. The mobile phase was 30% acetonitrile:70% water at 30oC.
- What is the mobile phase velocity?
- Find the retention factor k' for C6D6.
- Find the plate number and plate height for C6D6..
- Assuming that the peak widths for C6H5D and C6H6 are the same as that of C6D6., find the resolution for C6H5D and C6H6.
- Retention times for C6H5D and C6H6 are 193.3 and 194.3 respectively, Find the relative retention time and unadjusted relative retention time between C6H5D and C6H6.
Transcribed Image Text:Absorbance at 210 nm →→
Thiourea
(unretained)
t = 41.7 min
H₂N
S
NH2
188.1
D
min
D
D
D
4
5
D
W1/2=
1.01
min
2
Ал
At=
1.01
min
185
190
195
Time (min)
5
4
100
200
Time (min)
D
300
CD3
CD3 CH3
7
8
D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 1 steps

Recommended textbooks for you

