Q: A yellow balloon is filled with 0.5 mol of Ne and a red balloon is filled with 1.0 mol of Ar. Both…
A: Ideal gas equation given as PV = nRT P= pressure, V= volume, T= temprature n= moles, R = gas…
Q: A pure sample of Substance S is put into an evacuated flask. The flask is then heated steadily and…
A:
Q: 1. What is 1745mL stated in Liters?
A: We have been given 1745mL. mL represent millilitres. We have been asked to state 1745mL in litres.…
Q: A. Complete the table below. Use your knowledge on scientific notation and unit prefixes to complete…
A: Scientific notation is a way of expressing numbers in a more compact form. In scientific notation,…
Q: Consider the following reaction: Cl2O (g) + CO (g) → Cl2 (g) + CO2 (g) Initial rate of the…
A: Calculation of order of reaction: Let the order of the reaction with respect to CO is α. The rate…
Q: The pressure in a vessel that contained pure oxygen gas dropped at a rate of 0.440 atm/min as the…
A:
Q: 10. Compound A (s) is decomposed into B (g) and C (g). Equilibrium constant of this reaction is…
A:
Q: Calculate the root mean square (rms) average speed of the atoms in a sample of xenon gas at 0.15 atm…
A:
Q: 1. Osmotic pressure: We can model the nutrient transport system of a plant as a column of water that…
A: Given that, the osmotic pressure of a sugar solution is π = 1 atm. We have to calculate the molarity…
Q: What factors would contribute to increasing the overall average speed (µrms) of a gaseous sample? a.…
A: The root-mean-square (rms) velocity, often denoted as "u_rms," is a statistical measure of the speed…
Q: Name each of following compounds:
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: 2. Identify any missing formal charges on the following structures. H-C- -0-CH₂-CH₂ ww :0: H₂C-S-CH₂…
A: formal charge = number of valence electrons - bonding electrons2 - number of non bonding valence…
Q: Change An exothermic chemical reaction between a solid and a liquid results in gaseous products. A…
A: Answer: Spontaneity of the reaction can be decided on the basis of the value of Gibbs free energy…
Q: Nitrogen gas (N2) reacts with hydrogen gas (H₂) to form ammonia (NH3). At 200°C in a closed…
A:
Q: Clearly describe how you would prepare 25mL of a 0.15M stock solution of potassium chloride…
A:
Q: Given the following coordination compounds; Compound R=[Pt(NH3)4(OH)2]SO4 i. Compound S=…
A: Isomerism is the phenomenon where two or more chemical compounds have the same molecular formula but…
Q: + F₂ catalyst The above reactants would undergo a catalyst ,ل + H H The above reaction would be an…
A: We know the types of reaction; Substitution reaction :- The chemical reactions in which an atom or…
Q: I get using the Arrhenius equation but even after calculating the e-[82.64 KJ/mol)/(8.314 J/mol.K ×…
A: During putting the value to get the value of exponential part you have to put the value of…
Q: 4. Draw resonance structures for the following cation. Use arrows to show the electron movement. 5.…
A:
Q: Calculate all the concentrations in equilibrium in a 0.4M solution of magnesium hydroxide magnesium…
A: We have find out the concentrations in equilibrium in a 0.4M solution of magnesium hydroxide.
Q: The solubility of AgBr (silver bromide) and AgI (silver iodide) in pure water are 1.654×10-4 g.L-¹…
A: The equilibrium that exists between the undissolved AgBr(s) and the dissolved ions is AgBr(s) ⇔…
Q: An aqueous solution of hydrochloric acid is standardized by titration with a 0.150 M solution of…
A:
Q: This graph shows how the vapor pressure of three liquids varies with temperature: vapor pressure,…
A: The liquid having the highest vapour pressure at the same temperature is the most volatile. Boiling…
Q: 6) A particular reaction has the following rate constant at different temperatures: Temperature (°C)…
A: We now the Arrhenius equation for activation energy determination. ln(k2/k1) = Ea/R x (1/T1 - 1/T2)…
Q: A chemical engineer is studying the following reaction: 2 H₂S(g)+30₂(g) → 2 SO₂(g)+2H₂O(g) - At the…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: Predict the product of this organic reaction: || CH3-C-OH + CH₂-OH Δ P + H₂O Specifically, in the…
A: Given structure of reactant
Q: The following chemical reaction is used to synthesize a flavouring agent that has an aroma similar…
A: Esterification Reaction: When primary alcohol is treated with a carboxylic acid in the presence of…
Q: How much energy is required to raise the temperature of 13.1 grams of solid aluminum from 21.4 °C to…
A: Given data Mass of solid aluminum (m) = 13.1 g Initial temperature (T1) = 21.4 °C Final…
Q: Fill in the orbital energy diagram for the boron atom. E 2s 1s > 2p- V
A: Atomic No. Of Boron atom is 5 which implies it has a total of 5 electrons.As per the Aufbau rule,…
Q: The alkali metals react with the halogens to form ionic metal halides. What mass of potassium…
A: Given -> Volume of Cl2 = 5.10 L Pressure of Cl2 = 0.950 atm Temperature= 285.0K Weight of K =…
Q: Explain how soaps or surfactants decrease the surface tension of a liquid. How does the meniscus of…
A: Soaps & surfactants are compounds that lower the surface tension of liquids. They work by…
Q: Hexaamminechromium(III) ion, [Cr(NH3)]³+ is a metal complex containing six ammonia ligands i. ii.…
A: Using Valence Bond Theory, the hybridization state of [Cr(NH3)6]3+ can be determined by examining…
Q: A line in the Balmer series of the emission spectrum of atomic hydrogen is observed at a wavelength…
A: The Balmer series in the emission spectrum of atomic hydrogen is described by the equation:…
Q: 4. Select all that is considered spontaneous a. Cleaning your room b. A piece of bread molding after…
A: A spontaneous process is a process that occurs without the need for an external influence and…
Q: The operations involved in preparing a diluted solution from a more concentrated solution are shown.…
A: Given , Equation : 0.363(±0.004)M ×15.00 (±0.03)mL100.00(±0.08) mL
Q: 1A H 2A Li Be Na Mg 3B 4B 5B 6B 7B8B K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn 1B 2B 8A 3A 4A 5A 6A 7A He B…
A: The electronic configuration can be written by using three rules : Aufbau principle The Pauli's…
Q: 6. For each of the following organic compounds, draw a condensed structural diagram. A.…
A: There are mainly three ways to represent the structures of organic compounds- Expanded structure…
Q: Only 1.3x10-6 moles of lead sulfate PbSO4 can be dissolved in 100 mL of a solution of sodium sulfate…
A: The solubility product of PbSO4 is Ksp=[Pb2+] [SO42-]
Q: A certain reaction is first order in N₂ and second order in H₂. Use this information to complete the…
A:
Q: Rank the elements or compounds in the table below in decreasing order of their boiling points. That…
A:
Q: Fill in the orbital energy diagram for phosphorus. 3s 3p
A: given element = Phosphorus atomic number = 15 to draw energy diagram.
Q: A 1.25 L vessel at 23°C contains three gases: N2, H2, and O₂. The system contains 2.20 g of N₂,…
A: The given data is as follows: The volume of vessel (V)= 1.25 L Temperature (T) = 23…
Q: 4. Select all that is considered spontaneous a. Cleaning your room b. A piece of bread molding after…
A: A spontaneous process is a process that occurs without the need for an external influence and…
Q: Q2/A) Identify the characteristic signals the you would expect in the diagnostic region of an IR…
A: Structures of methyl salicylate and methyl benzoate : Structures of aniline and benzamide:
Q: Problem 3. The following solvents cannot be used in the preparation of organometallic reagents such…
A: Organometallic reagents are chemical compounds that contain a metal-carbon bond, where the carbon…
Q: Use the following information to answer Question 3. Organic Compounds 1. C₂H6 2. C₂H16 3. C₁₂H26 4.…
A: We are given molecular formula of 4 hydrocarbons and asked to classify them as directed.
Q: Write a detailed mechanism for the following reaction. Draw the structures of all the expected…
A: In this question, we will give reaction mechanism and also find out the products A, B and C In…
Q: 5. Draw the structure of the major product for each of the following reactions. CHO NO2 A A
A: The given two reactions are the examples of Diels-Alder reaction. Note: The dienophile with…
Q: The average rate of disappearance of ozone (O3) in the reaction 2O3(g) → 3O2(g) is found to be…
A: we have to calculate the rate of appearance of O2
Q: A chemical engineer is studying the two reactions shown in the table below. In each case, she fills…
A: Chemical reaction A. HCH3CO2 (l) → CH3OH (g) + CO (g) ∆H = 172 kJ ∆S = 543. JK and T = 69.0Cor T =…
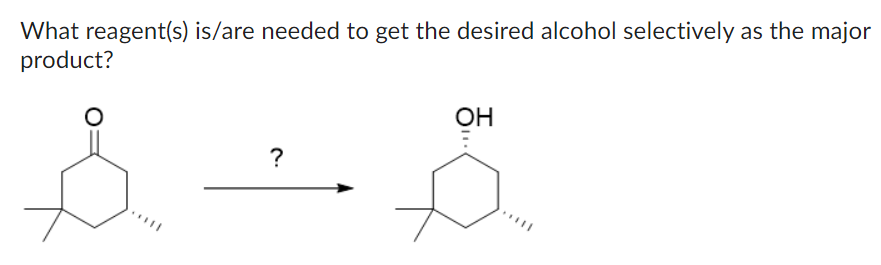
Step by step
Solved in 3 steps with 4 images

- What product is obtained from the reaction of each of the following alcohols with a. H2CrO4? b. HOCl? c. the regents required for a Swern oxidation? 1. 3-pentanol 2. 1-pentanol 3. 2-methyl-2-pentanol 4. 2,4-hexanediol 5. cyclohexanol 6. 1,4-butanediolhelp me draw the products when ethyl isopropyl ether is treated with the following reagent: 1.HBr (2 equivalents) 2.HI (in excess)Can you please check my answer for the following ochem question regarding the enols the the y-diketone CH3COCH2C(CH3)2COCH2CH3 can form. Thank you!
- 3) Which one(s) of the following is/are an acetal ?Which reagent(s) would accomplish the following reaction? 1) LiAlH4 and 2) H2O 1) CH3MgBr and 2) H2O H2O, H+ NaH NaOH, H2ODraw the structure of the product that is formed whent he compound 3-hydroxycyclohexanone undergoes a reaction with 1 equivalent of CH3MgI and then is treated with water. So normally the CH3 would attach and break the double bond with the O but this isn't the correct answer.
- What products would you expect from the oxidation of thefollowing compounds with (i) CrO3 in aqueous acid? (ii) withPCC?(a) tert-butanol(b) cyclohexanol(c) cyclohexanoneDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. [1] C6H5Li; [2] H2OWhat are the major products obtained when the following ether is heated with one equivalent of HI?
- Acetal product formation is difficult, since hemiacetals are stable while acetals are not stable. True FalseWill the bonding of the Zn2+ ion to the ketone oxygen lone pair make the ketone C=O easier or more difficult to reduce? WhySOCl2 (thionyl chloride) converts alcohols into alkyl chlorides. Explain How ?



