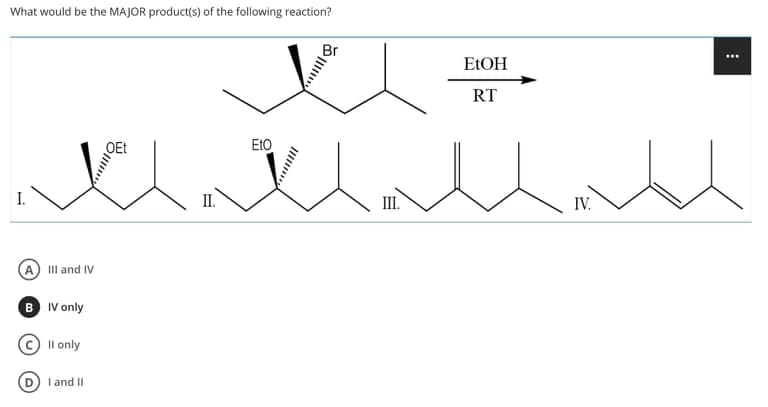
Q: Predict the main product of the reaction in Figure 10.
A: Alkyl bromide reacts with ammonia, a nucleophile to give a nucleophilic substitution reaction to…
Q: Predict all major and minor products. Show the intermediates and the movement of electrons as well…
A: (d) here, the alkyl is tertiary and tertiary alkyl halides with strong base (DBN) undergoes…
Q: Save Share II as starting mükerials show & synthusis that gets this produet benzene Cyelohexáura NO2
A: In this question, we want to synthesis the given Compound from the starting material. How we can…
Q: (E) An aprotic solvent is best for Sn1 reactions as they tend to help stabilize carbocation…
A:
Q: O A.D O B.A O C.E OD.C O E. B А + HCI - B с +
A: This question belongs to reaction mechanism branch of organic chemistry. Kinetically controlled…
Q: Which of the following is the most likely structure of the following carbocation after rearrangment…
A:
Q: Provide a plausible arrow pushing mechanism for the reaction below. Hint: NaOBr is in solution along…
A:
Q: How is this reaction is carry out ? Show all the steps please arrows and all the movement of…
A: The conversion of a primary alcohol to carboxylic acid via aldehyde is the oxidation reaction.
Q: 17. Provide all four products for steps i-iv. i. NaH, DMF ii. CH3CH₂Br iii. NaOH, H₂O iv. H₂O*, heat…
A:
Q: Carbonyl compounds as nucleophiles Maximum allowed tries per question: Unlimited No extensions will…
A:
Q: NH2 + CI HN Ho +
A: Given : We have to provide mechanism for the following reaction.
Q: Rank the following carbocations in order of stability. (The most stable is first.) II II O I > III >…
A:
Q: This reaction produces a predictable product. What is it? CH3 1. mCPBA 2. CH3 S Na
A: Given reaction,
Q: For next 2 problems, include a TS arrangement of hydroboration step that explains regioselectivity.…
A: When an unsymmetrical reagent is added to an unsymmetrical alkene then the negative part of the…
Q: 21) Carbocation forms in the reaction of: A) SN1 B) SN2 С) Both D) Cannot determined
A: We have to determine in which reaction carbocation is formed.
Q: Br e LOCH3
A: The solution is given below -
Q: using the starting material (on the left) to determine the sythetic route which will be the most…
A: Note : First disconnect the bonds. Then you will get the synthons. Start synthesis from 1-Ethyl…
Q: %3D arbocations in order of increasing stability (least stal cures may be equal in energy. II IV V
A:
Q: + Zn - 35 °C Ме-Li + Br + Mg
A: These are the general reaction: In 2nd reaction H is an acidic proton it lose H and gain…
Q: Which statement(s) is/are true regarding leaving Broups? L Weak bases make good leaving groups. II.…
A: I . ) Good leaving groups are weak bases. They're happy and stable on their own. Some examples of…
Q: 9. Follow the flow of electrons and show the intermediate and the final product. CH3
A: In number of reactions in organic chemistry , ring expansion is a quite important step to form a…
Q: The diagrams 1 to 3 below can be arranged to show the mechanism involved in a familiar reaction. The…
A:
Q: For each of the following reactions, predict the major product. 1. SOCI2 2. H2N (еxcess) HO, mCPBA…
A: 1) Reaction of carboxylic acid with SOCl2 produces acid chloride. Reaction of acid chloride with…
Q: For the reaction below: BH3 THF (solvent) a Draw the structure of the major organic product.
A: The above given reaction shows the hydroboration of alkene. Here, borane and hydrogen gets attached…
Q: Explain why I and II are not likely to be the major products of the following reaction. + AICI3
A: Friedel-Craft alkylation reaction:When the benzene reacts with an alkyl halide in presence of Lewis…
Q: b) Complete the following reactions. i. `NH2 „NH2 SOCI, i. iii. LIAIH4
A: In organic reaction, the reactants are organic compounds, the reaction mechanism depends upon the…
Q: Question 3
A: Organic compounds that contain a carbon atom directly bonded with metals such as magnesium, lithium…
Q: hoose a reagent from the table to bring about the following conversion: Br If more than one step is…
A: The Answer to the following question is-
Q: In each reaction box, place the best reagent and conditions from the list below. 1) 2) 3) 4)…
A: The given conversion can be done as follows : The first step is the alkylation of benzene ring which…
Q: Give the major organic product for the reaction paying particular attention to regioselectivity. "D…
A: See major products below.
Q: 2. Consider the following reaction called hydrovinylation from homework # 4: Clearly sketch out the…
A: Mechanism shown in step 2.
Q: Question 2 Draw all intermediates and the final product for the following reaction. Label all…
A:
Q: 1) a) Propose a detailed mec major product. Please include: 1) a perspective diagram of the…
A:
Q: Predict the major product of the reaction below. HNO, H,SO4
A: Interpretation - To predict the major product in the given reaction which is given in the question…
Q: Worksheet 9 – Chapter 8 | Given the information below, write out a reasonable mechanism for the…
A:
Q: d) Brg, FeBra e) F. Ph. Ph AICla IZ
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: SYNTHESIS PORTFOLIO (PART I) Synthesis of 1 2-epoxycyclopentane I. MATERIALS CAS # or Catalogue #…
A:
Q: Provide the structure of the major organic product in the reaction below. NBS Br Br A. Br C D Br O A…
A: 1. NBS = N-Bromosuccinimide, which is source of Br•. Alkene with NBS gives allyl bromine as major…
Q: Provide the major organic product in the reaction below. CH,CH2 + NaOCH, CH
A:
Q: 1. BH3 b) 3,3-dimethyl -1-butene -> 2. H2O2, OH
A:
Q: For the reaction given below, predict which product will be the major one and by what mechanism it…
A: In given reaction - Sovlent used is DMF which is aprotic and non polar. In reactant —OTs is very…
Q: product formed when the compound shown below undergoes a reaction with 1 equivalent of AcCI. ACCI =…
A: Using concept of nucleophilic substitution by N- center.
Q: that are, draw the products. For those that are not, explain why. (Hint: Ex nvolve drawing the…
A: The question is based on the concept of mechanisms. we have to identify the error in the following…
Q: OH3C HCO- 1 hoi H 2 1) C4HgLi 2) THF OH3C ΗΤΗ H OIS H 3
A:
Q: How would you make the following compound? Show all intermediates and reactants. Also, how would the…
A: Sodiummethoxide (NaOMe) reacts with alkyl halide forms alkene, which undergoes NaIO4 in presence of…
Q: When hydroxide is used as the base to carry out an E2 reaction on a vinylic halide, the reaction…
A: E-isomer: In an alkene if both the group along double bond is in opposite side is called E-isomer.…
Q: i) NaNO2, HCI ii) H2O, heat NH2 (e) H3C NaNH2 NH3 (1) (f)
A: Major products of following organic reaction
Step by step
Solved in 2 steps with 1 images

- Fill in necessary products reactants or reagants of these reactions. Please note the existence of enantionmers in some cases.What is (are) the elimination product(s) of the following reaction? Only I Only II Only III II and IIIWhat is the organic chemistry mechanisms to get to this final product? one reagant is NaOCH2CH3
- Predict the main product of the reaction in Figure 10. * A B C D The main product of the reaction cannot be predicted.How to determine the type of reaction? (SN1/SN2/elimination)?Arrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, I
- In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:What would the final products look like? Pls specify stereochemistry if neededChemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?
- Choose the best reagents from the list provided below for carrying out the following conversion. Match the reagent with the step number. HCl (aq), Zn(Hg) KMnO4, H3O+ CH3Cl, AlCl3 HNO3, H2SO4 Cl2, FeCl3 fuming sulfuric acidPredict the product(s) of the reaction below: Al₂O3 → which is it below O2 AI AI + O AI +O2Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?

