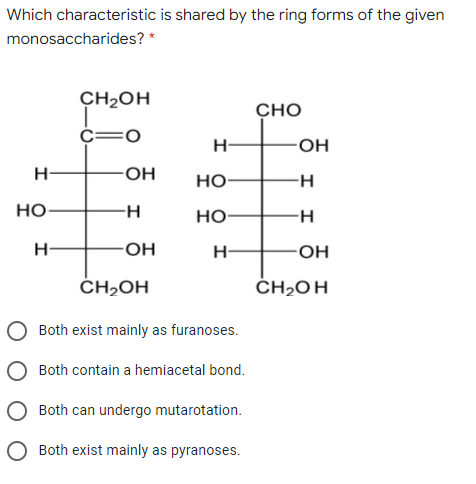
Which characteristic is shared by the ring forms of the given monosaccharides? * CH2OH CHO O= H- HO- HO- но- H- но- Но H H- -OH ČH,OH ČH2OH Both exist mainly as furanoses. Both contain a hemiacetal bond. Both can undergo mutarotation. O Both exist mainly as pyranoses.
Q: What compounds remain in the ethanol solution after the DNA is removed bt spooling onto a glass rod?
A: Introduction: The extraction of DNA is the first step for subsequent molecular or forensic…
Q: In Figure 4C, we see the inhibition of LRRK2 kinase activity by nanobody 1 (Nb1). Using the…
A: The Lineweaver-Burk plot is used for calculating the Vmax and the Km values in the enzyme kinetics…
Q: he eukaryotic metallothionein gene promoter consists of all EXCEPT: MRE GC Box Pribnow box…
A: Metallothioneins are proteins that have high affinity towards binding heavy metal ions, and its…
Q: . This cell is dependent solely on glucose as an energy source a.Muscle cells b.Kidney cells…
A: 1. the cell that solely dependent on glucose source is- Answer- d. brain cells Glucose in human…
Q: Ön average, 180 liters of plasma are filtered each day. A If humans had to expend one molecule of…
A: Introduction: A mole of any substance contains as many elementary units (atoms and molecules) as the…
Q: Lec 3 Protein Digestion Make a flow chart of: (Indicate the organs, enzymes and how it functions)…
A: In process of digestion, complex molecules are converted to simple molecules with the…
Q: Benefits of a project "Drinking Water Refilling Station " to individuals and families and community…
A: Refilling stations for water bottles are now one of the most popular methods of decrease the…
Q: Key features of signal transduction mechanisms are: specificity, amplification, integration and…
A: Receptors are present in the body. The drug molecules are bound to such receptors and show their…
Q: Categorize the electron carrier by the number of hydrogens that it can transfer in a single…
A: In the process of electron transport, one or more molecules accept electrons from another and donate…
Q: fat-soluble vitamins?
A: Fat soluble vitamins:-These vitamins are present in foods containing fats,the body absorbs these…
Q: Outline how ammonia is generated and detoxified, and how carbon skeletons are further processed,…
A: Ammonia is a colourless, odourless gas composed of hydrogen and nitrogen. Ammonia is required for…
Q: Differentiate the difference between saturated fat and unsaturated fat. Explain the chemical…
A: Fatty acids are important micromolecules which combine together to form lipids in plants, animals…
Q: discuss the biosafety issues involved in the use of genetically engineered microorganism.
A: Biotechnology or genetic engineering where organisms including plants, animals and bacteria or…
Q: Retroviral delivery of the gene coding for Combined Immunodeficiency. treats individuals with Severe…
A: Successful gene therapy that is performed with the retroviral vectors neceserily requires robust…
Q: What amino acid side chains can be modified by methyl groups? What is unusual about methyl group…
A: What amino acid side chains can be modified by methyl groups? Answer: Methylation is a process of…
Q: 1. A student, halfan hour after the dinner, containing about 150 g of carbohydrates, 20 g of fat,…
A: Fats are glycerides (mono, di and tri), sterols, phospholipids and free fatty acids. it is stored in…
Q: a. Is the disaccharide below a non-reducing sugar? yes or no b. The glycosidic linkage in the…
A: Carbohydrates are composed of carbon, oxygen, and hydrogen which are connected by the…
Q: A glycosidic bond between two monosaccharides can also be classified as a(n) ________
A: Monosaccharides are the basic sugar unit of carbohydrates compounds. Monosaccharides which cannot be…
Q: At higher amounts of protein, the Bradford assay is not linear. Consider the plot to the right: what…
A: Bradford assay is used to determine the unknown concentration of proteinbin a sample from standard…
Q: Which of the following compounds is derived from arachidonic acid with the help of the enzyme…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: The reaction is reversible. Check all that apply. acetyl CoA lactate ethanol
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: How does the degree of unsaturation and structure of fats affects its functionality, for example in…
A: Lipids are not polymers. The simplest form of lipid is fatty acids which are a long chain of…
Q: Match the following methods of Analysis Fractional analysis, methylation, and periodate oxidation A.…
A: Methods of analysis are different techniques used to analyse a compound for determination of its…
Q: The conversion of glyceraldehyde-3-phosphate to 1,3-bisphosphoglycerate is the only oxidative…
A: Glycolysis is a metabolic pathway in which glucose is converted into pyruvic acid and ATP releases.…
Q: Sphingolipids do _________________. I. contain a glycerol core with a phosphocholine…
A: Glycerophospholipids and sphingolipids are complex lipids. They're present in biological membranes…
Q: How many ATP couny after complete oxidation 1 mol glucose
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic…
Q: VO (mM/min), Minus Substrate, mM vO (mM/min), Plus Inhibitor Inhibitor 0.5 23.5 16.67 1 32.2 25.25…
A: The reciprocal of the given kinetic data is as follows: 1/[S] mM-1 1/V (min/mM) minus inhibitor…
Q: 1. In human diet, all cholesterol comes from Animal products Vegetable products Both A and B None of…
A: "Since you have posted multiple question we will answer the first question for you. If you want any…
Q: Which of the following reactions is the most exergonic? a Conversion of PEP to Pyruvate b…
A: Exergonic reaction is the reaction in which energy is released when reactants are converted to…
Q: Explain the roles of transamination, oxidative deamination and the urea cycle in amino acid…
A: Transamination and deamination are two very important processes of amino acid degradation. The…
Q: Blood type O is also known as universal donor because it does not contain plasma antibodies. True…
A: Depending on the presence or absence of antigen or antibodies, the blood has been categorised into…
Q: Fill up the table by giving examples of common food, their pH range and classification (acidic…
A: The food items we consume are of different pH ranges. The acidic food items are responsible for…
Q: Equivalence in ATP Products from glycolysis 1. 2. ATPS produced АТР ATP ATP NADH ATPS 3. Pyruvate…
A: Net equation of Glycolysis : glucose + 2 NAD+ + 2 ADP + 2Pi ⇒ 2 pyruvate + 2 ATP + 2 NADH…
Q: Which isomer- (R)-arginine or (S)-arginine is D-arginine? O (R)-arginine O (S)-arginine Submit…
A: Isomers are molecules with same molecular formula and different arrangement of atoms. Enantiomers…
Q: Which of the following statements are TRUE? O a. Phosphofructose kinase (PFK) is used exclusively…
A: Carbohydrates are made up of carbon, hydrogen, and oxygen in the ratio of 1:2:1. Carbohydrates act…
Q: Fibrous proteins, globular proteins, and conjugated proteins are the three (3) primary kinds of…
A: Proteins which are most essential part of diet they are building block of body . Protein named by…
Q: Which undergoes more number of beta-oxidation cycles - degrading a 16:0 lipid or degrading a 16:2…
A: β-oxidation is the process of degradation of fatty acids by the oxidation of βcarbon atoms of the…
Q: 4. Draw the condensed structural formula for the triacylglycerol made from 3 saturated fatty acids…
A: Triglycerides are the main constituents of body fat in humans and other vertebrates and are also…
Q: 1.. Discuss how the inability to synthesize insulin and glucagon would impact lipid and/or…
A: Insulin is a hormone produced by beta cells of pancreas that maintains the levels of sugar in the…
Q: How many ATP and NADH molecules would be produced during the citric acid cycle if 3 molecules of…
A: Cells are the basic unit of life. Cells derive energy from the metabolism of biomolecules such as…
Q: In bacteria, single polycistronic mRNA encodes for: a single mRNA which is split into…
A: Polycistronic mRNAs are those that code for many polypeptides from a single mRNA as it involves…
Q: DATA TABLE Part I Specific Rotation of Sugars Angle blank (°): 71.8 Height (cm) Angle (*) Specific…
A: Every substance with the ability to rotate a plane polarized monochromatic light will have a…
Q: What is the role of decarboxylation in fatty acid synthesis? Describe another process discussed in…
A: Fatty acids are the body's fat-building blocks. Acetyl-CoA is used to make fatty acids. Fatty acid…
Q: What are the risks (factors that can lead to the disease) and challenges (side effects of…
A: Cholesterol is a waxy molecule found in the blood stream.. High cholesterol levels can put you at…
Q: 3. A biochemist purified a protein from the bloodstream and determined its molecular weight under…
A: The quaternary structure of a protein describes the association of polypeptide chains or subunits…
Q: To establish a standard curve for a BSA standard curve using Bradford, the spectrophotometer…
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. These…
Q: The fact that some eukaryotic rRNAs are self-splicing indicates that RNA structures are highly…
A: Introns are non-coding sequences, which are removed during the process of splicing. Splicing is…
Q: Entire sequence below needs to be amplified by PCR and subcloned into a plasmid vector. Which of the…
A: PCR, or the polymerase chain reaction, is a technique used by molecular biologists to amplify…
Q: 1. A student, halfan hour after the dinner, containing about 150 g of carbohydrates, 20 g of fat,…
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since…
Q: A glucometer measures the electrical current produced by the redox reaction catalyzed by glucose…
A: Introduction: The level of blood glucose is normally maintained within a narrow range under various…
Step by step
Solved in 3 steps

- In the following list, identify the carbohydrate, the fatty acid, the amino acid, and the polypeptide: a. NH2CHRCOOH b. C6H12O6 c. (methionine)20 d. CH3(CH2)16COOHWhich linear monosaccharide below corresponds to this cyclic one? A) A B) B C) C D) D E) None of themWhy are polysaccharides (e.g. starch) not soluble in cold water but are soluble in warm water?
- Which two monosaccharides make up this disaccharide? A. Two alpha mannoses B. Two alpha glucoses C. Two alpha galactoses D. One alpha mannose, one alpha glucose E. One alpha mannose, one alpha galactose F. One alpha galactose, one alpha glucose G. One beta mannose, one alpha glucose H. One alpha mannose, one beta glucoseStarch can be separated into two principal polysaccharides, amylose and amylopectin. What is the major difference in structure between the two?Which of the following statements about monosaccharides is true? A. Monosaccharides are classified according to the location of their carbonyl group. B. The penultimate chiral center of a monosaccharide determines its handedness. C. If the carbonyl group of a monosaccharide is terminal then it will react positively with Barfoed's and Seliwanoff's tests. D. The cyclization of monosaccharides results to the formation of a cyclic ether. E. The existence of anomers is due to the mutarotation along the reference carbon
- Plants often produce waxes on the surface of their leaves toprevent dehydration and protect against insects. What structural feature of waxes makes them more suitable for this taskthan carbohydrates or proteins?Draw the ketose and aldose forms of a monosaccharidewith the chemical formula C3H6O3. How is the structureof the monosaccharide changed from one form to theother in the human body?When blood levels of glucose are higher than normal,glucose molecules react with protein side chains such as thatof lysine residues to produced glycated side chains. Explainwhy this process is dangerous. What is the name of the firstfunctional group that is formed during glycation?



