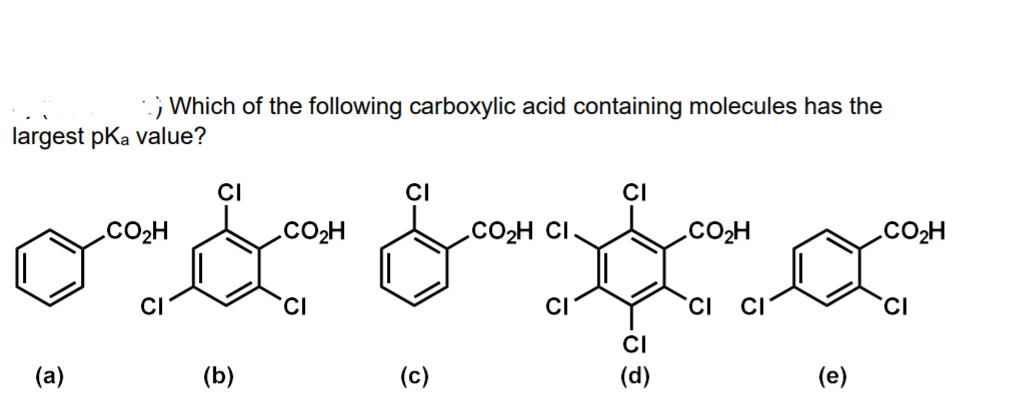
;; Which of the following carboxylic acid containing molecules has the largest pKa value? CI CI CI CO2H .CO2H .CO2H CI CO2H .CO2H CI CI CI' CI (d) (a) (b) (c) (e)
Q: Explain why aniline (on the left; pK, = 9.13) is a stronger base than cyclohexylamine (on the right;…
A: Amines are basic in nature due to their ability to donate lone pair of electrons present on…
Q: The weakest of the acids below is and its pK is . The strongest of the acids below is |and its pK is…
A: Pk value - which measure the strength of acid in logarithmic scale.
Q: mula C13H100 . Soluble in H2SO4 ve a trace. Compounds (A) give negative result with chromic acid
A:
Q: Describe the trends in the acidityand physical properties of carboxylicacids, and explain how their…
A: Acidity of Carboxylic acids is based on the stability of the carboxylate ion. It is higher than…
Q: 11. Identify compounds A and B in the scheme below. OMs DBU (bulky base) Ph Boo Compound A? Compound…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: ver to the ammonia on warming with dilute aqueðus sodium hydroxide, X could be A) ethanamide B)…
A:
Q: cOOEt -014 resovcinol ethyl accteacebate methylumbelliferone 04 conduyote acid . -0lt Ho. -OH olt Ho…
A: Chem sketch of given diagram has to be given,
Q: Treatment of indene with NaNH2 forms its conjugate base in a Brønsted–Lowry acid–base reaction. Draw…
A:
Q: What are the pKa’sof HCN (hydrocyanic acid), and HF (hydrofluoric acid)? Based only on those…
A: Pka of HCN is 9.2 Pka of HF is 3.2 From this data we can say that HF is more acidic than HCN. This…
Q: }) 2) но 0 هر n Ow + H ala + on olf Ome [H+] -4₂0 MeONa MeoNa меон
A: An organic reaction generally involves addition reaction, substitution reaction or elimination…
Q: ety and labl the Lewis acid (electrophile) nd the Lewis base (mcleophile) in the fllowing actions…
A:
Q: When diphenyl ether is reacted under the same conditions as in Problem 22.35, a compound is produced…
A:
Q: Rank the reactivity of the following carboxylic acid derivatives towards hydrolysis. Assign '1' for…
A: The answer is given as follows
Q: Mol of 5-hydroxypentanoic acid reacts with 1 mol of SOCI2 and then with 1 mol of dimethylamine. What…
A: In a chemical reaction, two or more compounds react with each other to form product(s). Organic…
Q: 4. reactivity towards hydrolysis HO. NH2
A: In this question, we will see the reactivity order ( for least Reactive 1 and for most reactivity…
Q: To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? A (1)…
A: Sceheme 1: Step 1 : propanol reaction with PBr3 results bromo propane. Step 2 : Bromo propane…
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A:
Q: Which of the following will give a negative result with Br2/CCI4? O a) O b) O d) H. Which of the…
A: Br2 in the solvent of CCl4 give addition reaction on alkene and alkyne ( called trans addition…
Q: Rank the fallowmg benzoic acid denvatnues frm weatest Clisted frst) to Člasted last): stringest -olt…
A: We have four compounds , we have to predict their acidity order.
Q: 4. Calculate the ratio of the concentrations of a thiolate (A) and its corresponding thiol (HA) at a…
A: a) Given : pH of solution of HA = 5.5 And pKa of HA = 10.3 => Ka = 10-pKa = 10-10.3 = 5.012 X…
Q: Show the steps necessany to transform the Compound o the left into the acid on the Might,…
A:
Q: Rank the attached compounds in order of increasing acidity.
A: Acidity of any molecule depends on the stability of the anion formed after removing the most acidic…
Q: 5. Formic acid (HCOOH) has a pka of 3.75. (a) What percent of formic acid dissociates at pH 4?
A: Given, pKa of formic acid=3.75
Q: To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? a)…
A: Scheme 1: Step 1 : propanol is converted into bromo propane using PBr3. Step 2: Bromo propane is…
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: Species with higher pKa will be more basic. In contrast, species with lower pKa will be stronger…
Q: Silicic Acid structure is this: The pK,s are: 9.83 and 13.1 он но - si- он OH Draw each acid…
A: a.) Given compound is a weak acid with high values of pKa. To write acid reactions , we show it…
Q: The pkb of methylamine, CH3NH2, is 3.36. Calculate the pk, of its conjugate acid, CH3NH3*. pka =
A:
Q: Which is true regarding the direction of the following reaction? CH3COOH (aq) + H2PO-4 >> CH3COO- +…
A:
Q: (a) Que the albuciones of missing heats raks (ख) and products & thandl Pentanal Acetic acid Salicy…
A: Here we are required to draw the structure of the ester formed.
Q: carcinogenic properties. Four students attempted to synthesize the dye using different starting…
A: Synthesis of Dye:
Q: draw the structure of vanillin acetate with all protons
A:
Q: Which mixture can be converted into diastereomeric mixture when reacted with the following base: CH,…
A: Diastereomers are optically active species having same absolute configuration at one centre whereas…
Q: 2. Consider the two cydlic molecules below A. B. a. The CH2 hydrogens of molecule A. and B. show…
A:
Q: Rank the following compounds in order of increasing basicity: 1.p-nitroaniline I p-aminobenzaldehyde…
A:
Q: Explain the trends in the acidity of phenol and the monofluoro derivatives of phenol. OH он он он F…
A: Higher the pKa lower is the acidity. If the conjugate base is stabilized, then it is more acidic.
Q: Explain this result: Acetic acid (CH3COOH), labeled at its OH oxygen with the uncommon 18O isotope…
A: It is given that acetic acid is reacting with a base NaOH and is undergoing acidification and the…
Q: Predict the major product(s) when propanoic anhydride is reacted with: a. MezNH (excess) b. Isobutyl…
A: Propanoic anhydride a. when propanoic anhydride is reacted with dimethylamine(Me2NH) dimethyl…
Q: Rank the following compounds in order of increasing basicity: CH3O- , H2N- , H2O and NH3
A:
Q: Using the pk, table, estimate pK, values for the most acidic group on the compounds below, and draw…
A:
Q: 3) . Which of the following carboxylic acid containing molecules has the largest pKa value? CI ÇI CI…
A: Concepts of acids and bases.
Q: Please rank the following by increasing acidity: H2Se, H2O, CH4, H2S
A: Step → 1Fow key factors that affectacidic. Nature :-(1) Atom: - acidity increases in any compound as…
Q: Consider the Struchurs of pyrolidine and N- nuthylpyrolidine belaw: CH3 Purrolidine…
A:
Q: Histidine is an important catalytic residue found at the active sites of many enzymes. In many…
A: Histidine is the substituted alanine and is aromatic imidazole. This aromatic ring structure will be…
Q: 3. According to the Evan's pKa table, phenol has a pka of 9.95. p-Nitro and m-nitrophenol have pKas…
A: Ka is the acid dissociation constant. pKa is defined as the negative logarithm to base 10 of Ka.…
Q: The pK, and pK,2 of ortho and para isomers of phthalic acid (diprotic acid) are shown below: pK pK2…
A: There are number of factors detaining the acidity of benzoic acid. These are, 1. Inductive effect…
Q: Aniline C6H5NH2 has a base dissociation constant (Kb) of 4.3 × 10-10. What is the conjugate acid of…
A:
Q: What percent of acetic acid is present in the acidic form at pH 5.0, assuming a pKa of 4.8?
A: Given,pH = 5.0pka = 4.8
Q: Solve the Attachment
A: Acids tend to lose H+ and form conjugate base. Stronger the acid, more is the Ka value and smaller…
Step by step
Solved in 3 steps with 1 images

- The pKa value of CH3COOH/CH3COO- is 4.75. At what pH value are the concentration of CH3COO- and CH3COOH the same?At what pH will 25% of a compound with a pKa of 5.3 be in its basic form?a. For this acid-base reaction: C⊖H3 + H3O+ −−→ CH4 + H2O, the conjugate acid of carboanion (C⊖H3) is _________________ b. Complete the equation: HC≡N + NaOH −−→ ___________+__________ c. Rank the acidity of these: NH3, HF, H2O, HCl, NaH Most acidic______>_______>_______>_______>_______least acidic d. The Ka of acetic acid (found in vinega) is 1.8×10-5. Then the pKa of formic acid is
- Nitric acid (HNO3) is a strong acid that completely ionizes when dissolved in water (pKa = -1.30). Calculate the pH of 10 mg/L HNO3. a. Unlike nitric acid, butyric acid (CH3CH2CH2COOH) is a weak acid (pKa = 4.8). Calculate the pH of a solution when 90% of the CH3CH2CH2COOH is dissociated. b. Briefly explain how you can use the pKa values of these two molecules to determine that nitric acid is a strong acid, while butyric acid is a weak acid.Acetic, formic, hydrofluoric, and acetylsalicylic acids have the following values of pKa (25°C), respectively: 4.74, 3.74, 3.17, 3.49. Order these acids in order acidity (from highest to lowest acidity)Hypoglycin A, an amino acid derivative found in unripened lychee, is anacutely toxic compound that produces seizures, coma, and sometimesdeath in undernourished children when ingested on an empty stomach. (a) Draw the neutral, positively charged, and negatively charged forms of hypoglycin A. (b) Which form predominates at pH = 1, 6, and 11? (c) What is the structure of hypoclycin A at its isoelectric point?
- The side chain of acetyl-L-lysine-OMe has a pKa=10.5 and its acid-dissociation reaction is shown below. C. what is the pH of a solution wherein the ration of [A-]/[HA]=1:10?Can any functional groups on Risperidone ionize? If so, which ones? Under what conditions? What are the pKa values of these functional groups? Write the structures in their correct ionization state at physiological pH.Carbonic acid is a weak di-protic acid, which constitutes an important buffer system in the blood. There, it can dissociate two different protons at two different pka’s. These pka values are 6.4 and 10.2 a) Research the chemistry of this system in your textbook and provide the balanced equilibrium reactions. Start with the formation of carbonic acid from carbondioxide and water and show the reactions where protons dissociate. For each step provide both the chemical formulas and the names of the chemical components. Indicate the above pka values for the appropriate equilibrium reactions b) Provide both the chemical formulas and the names of all molecules shown in a) that can act as i) weak acid and ii) conjugate base at the respective pka values
- The side chain of acetyl-L-lysine-OMe has a pKa=10.5 and its acid-dissociation reaction is shown below. A.. for this reaction, which form of lysine represents A- and which is HA?Experimental observations show that thiocyanate (SCN- ), the conjugate base of thiocyanic acid (pKa = 1.1 at 25 °C), is quite soluble in neutral water at pH > 2. In the space provided below, explain this experimental observation. That is, why does SCN– readily dissolve in water when the pH > 2?The pKas of chemicals HX and HY are 5 and 7 respectively. The pKa of carbonic acid H2CO3is 6. If you made up an ether solution of chemicals HX and HY in a separatory funnel, andthen added an aqueous solution of sodium bicarbonate NaHCO3 to that separatory funnel,would both HX and HY stay in the ether layer? Or would either or both of them transfer intothe aqueous layer? If one goes into the water layer, will it be in it’s neutral HX/HY form, or init’s deprotonated anionic form?

