Q: Nitration of benzene is carried out by which of the following reactive ?species NO3 O HNO3 O "NO2 O…
A:
Q: c) Propose structures for A- C in the following reactions: Free radical initiator RCH=CH2 NaHSO3 A…
A: Anti-Markovnikov rule describes the regiochemistry where the substituent is bonded to a less…
Q: Write the appropriate reagents, conditions and products for the following electrophilic aromatic…
A: In this question we have to tell the product of the reaction.
Q: Which type of reaction – addition, elimination, rearrangement, substitution,- best describes each of…
A:
Q: 5- Show radical chain process for the following reaction (Explain in details with mechanism !) onit…
A: Free radical reaction occurs in the presence of light to form free radical and these radical take…
Q: Hydration of alkynes is performed in the presence of Hg2+* salts but hydration of alkenes can be…
A: why an acid catalyzed used in hydration of alkenes reaction- Acid catalyzed hydration of alkenes…
Q: Does the data provide evidence for the claim that the reaction of Br2 with alkenes proceeds via…
A: Note: trans stilbene + trans addition (bromine addition) results 1,2-dibromo-1,2-diphenylethane.
Q: The correct decreasing order of reactivity toward nucleophilic addition reactions is CHO H. II III…
A:
Q: Aromatics can be converted into nitroaromatics upon treatment with a mixture of nitric and sulfuric…
A:
Q: Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Draw…
A:
Q: مع مسن NO₂ NaCN CN NO₂ Although aromatic substitution reactions usually occur by an electrophilic…
A: The rule for electron flow is that electron flows from electron rich center or bond to electron…
Q: Which set of conditions for the reaction given below would provide the largest amounts of…
A: We have to find a suitable reagent that will give the largest amount of substitution product.
Q: CHE2613/101/0/2022 3.2 The reaction of methylpropene with HBr, under radical conditions, gives bve…
A: Free radical takes place when homolysis cleavage takes place.
Q: Q1: B: How does alkylation with sulphuric acid and hydrofluoric acid work, and what are the…
A: The alkylation process is the process that combines light iso-paraffins, mostly isobutane C4H10…
Q: In an attempt to prepare propylbenzene, a chemist alkylated benzene with 1-chloropropane and…
A: Friedel-Craft alkylation reaction: When the benzene reacts with an alkyl halide in presence of Lewis…
Q: CF2Cl2 can be prepared from the nucleophilic substitution of another alkyl halide CCl4 in the…
A: Given-> CCl4 + 2HF ------> CF2Cl2 + 2HCl. Rate = K[CCl4] Rate constant (K) = 1.5 × 10-4 s-1…
Q: ihe diagram below shows two synthetic pathways by which the compound W might be prepared. C3H;OH…
A: Stepwise reaction upto W from first path Here first we chose oxidising agents and then followed by…
Q: divls depends oin the moisture content of the reaction mixture. Propose a detailed mechanism for…
A:
Q: Br H3C H3C CH2 cyl halides undergo nucleophilic substitution and elimination reactions. When the…
A:
Q: Choose the most stable intermediate in the nucleophilic substitution aromatic (SNAR) reaction of…
A:
Q: Explain the steps involve in the below organic reaction in detail ONa H3N
A:
Q: Generally alkynes prefer to undergo: SN2 Both SNI and SN2 SNI Addition Reaction
A: Alkynes are unsaturated system (due to the presence of multiple bonds in them). So, their principle…
Q: 3. How many regioisomeric products (including minor products) can form in radical substitution…
A: Interpretation: We have to draw the Regio isomeric products.
Q: What is the reaction mechanism? Scheme 30. Intermolecular Diastereoselective Allylic Ammonium Ylide…
A:
Q: Draw the structure(s) of the organic product(s) predicted when this compound reacts with NABH4,…
A: NaBH4 acts as a reducing agent in organic reactions. In presence of carbonyl groups, the BH4-…
Q: Benzene reacts with CH3CH2CI in the presence of AICI3 to form ethylbenzene (Friedel- Crafts…
A:
Q: Which of the following should not be included in a synthetic scheme? Reaction temperature Reaction…
A: A synthetic scheme is a process where simple or commercially available compounds are converted into…
Q: C(CH3)3X +H20 C(CH3)3OH where (X-F, Cl, Br, I) give reaction with each of halogen as well determine…
A: An organic problem based on SN1 mechanism, which is to be accomplished.
Q: Br CF3 CH3 OCH3 6888 b C d
A:
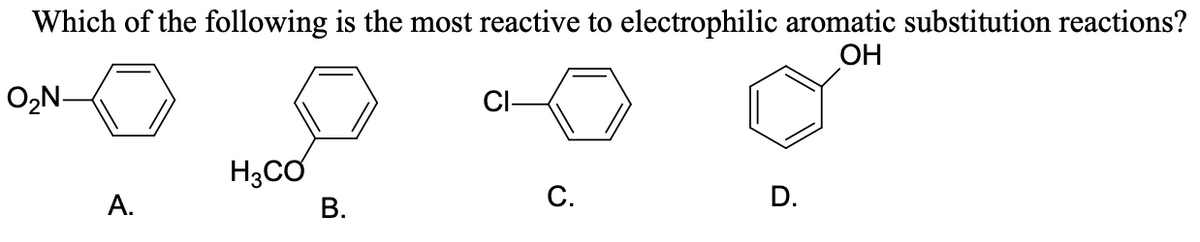
Q: CoHSOH, C6H5SO3H, CgH5Cl
A: In electrophile aromatic substitution reaction, an electrophile (E+) substitutes hydrogen atom from…
Q: Give the major organic product(s) of the following reaction. HO OMe ? + PCC Meo OMe
A: SOLUTION: Step 1: PCC (Pyridinium chlorochromate) is a mild oxidizing agent. It converts the primary…
Q: 38) Which of the statement/s below is/are not correct? a. Nucleophilic substitution reactions…
A: A multiple choice question based on nucleophilic substitution reactions, which is to be…
Q: . Terminal alkynes react with Br2 and water to yield bromo ketones. For example: `CH;Br Propose a…
A:
Q: Does the data provide evidence for the claim that the reaction of Br2 with alkenes proceeds via…
A: Note: trans stilbene reacts with bromine results 1,2-di bromo - 1,2-diphenyl ethane confirms anti…
Q: Name the following organic compounds. For full credit, indicate whether each of the alkenes is E or…
A: Representation of geometrical isomers is done via two specified symbols E and Z. In this notation,…
Q: Question attached
A:
Q: Account for the following relative rates of solvolysis under experimental conditions favoring S,1…
A: Concept introduction: SN1 reaction: The displacement of atom or group by nucleophilic is known as…
Q: L3 Fit to page O Page view A) 7. (i) What would you expect to be the order of reactivity from the…
A: Electrophilic reaction will be favorable at that position where elctron density is high.
Q: Heterocyclic compounds plays an important role in our daily life. They are mainly used in…
A: The structure of thiophene: A halogen group can reacts with thiophene to form a suitable compound.…
Q: Answer ALL parts of this question (a) Electrophilic substitution reactions of benzene are used to…
A:
Q: In Nicolaou's total synthesis of taxol (Ref.: J. Am. Chem. Soc. 1995, 117, 653) the carbonyl group…
A: This is an example of conversion of ketone to vinyl Lithium via tosylhydrazone intermediate.
Q: For the reaction given below, predict which product will be the major one and by what mechanism it…
A: In given reaction - Sovlent used is DMF which is aprotic and non polar. In reactant —OTs is very…
Q: Q6
A: Chlorination of alkenes in presence of UV light undergoes substitution reaction. The reaction…
Q: The following Friedel-Crafts Alkylation reaction usually results from the disubstituted product from…
A:
Q: Which type of reaction – addition, elimination, rearrangement, substitution,- best describes each of…
A:
Q: V(CO)6 undergoes CO substitution by an associative mechanism. explain and show the complete…
A:
Q: The reaction of a phosphonium ylide with a ketone or aldehyde produces an alkene, but, as shown in…
A: Witting reaction is the reaction between an aldehyde or ketone with a ylide. A ylide is a neutral…
Q: Five monosubstituted benzenes are represented below. List the above compounds in order of increasing…
A: Reactivity towards aromatic electrophilic substitution reaction.
Q: In Nicolaou's total synthesis of taxol (Ref.: J. Am. Chem. Soc. 1995, 117, 653) the carbonyl group…
A: In this question, we will discuss about the mechanism and then give the name of the Reaction. You…
Step by step
Solved in 3 steps with 3 images

- In SN2 reactions of haloalkanes, the order of reactivity is RI>RBr>RCl>RF. Alkyl iodides are considerably more reactive than alkyl fluorides, often by factors as great as 106. All 1-halo-2,4-dinitrobenzenes, however, react at approximately the same rate in nucleophilic aromatic substitutions. Account for this difference in relative reactivities.Dr@w a carbene and explain how it can function as a nucleophile or electrophile.17. Which aromatic compound in figure 17 will be least reactive in an electrophyllic aromatic substitution reaction?
- Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with isobutylene and HFElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with HNO3, H2SO4.Which of the following is the major organic product in the reaction sequence in Image 21? A. b B. c C. a D. d
- Compound name is Toluene . Name the structures of all possible chemical (Electrophilic aromatic substitution) reactions of the compound namely; a. Halogenation (Chlorination or Bromination) b. Nitration c. Sulphonation d. Friedal Craft Alkylation e. Friedai need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?
- H8. Organic chemistry Text answers and (descriptive structures and / or reaction mechanisms). Explain the chemistry of the carbonyl group (direct addition of nucleophiles and addition with elimination)What are the steps of the mechanism of reaction . Electrophilic aromatic substitution. Is it Ortho,para,meta ?In electrophilic aromatic substitution reaction by drawing the structures of the compounds given below Explain which products will be formed as a result of the reaction with the nitronium ion.a)Methoxybenzeneb)Benzoic acid

