Which of the substances in question 12 releases less energy per gram on complete combustion to CO2 and H2O? O Hexanoic acid glucose O Both release the same amount of energy as they are in equilibrium O Cannot be determined
Which of the substances in question 12 releases less energy per gram on complete combustion to CO2 and H2O? O Hexanoic acid glucose O Both release the same amount of energy as they are in equilibrium O Cannot be determined
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter6: Basic Clinical Chemistry
Section6.5: Blood Glucose And Hemoglobin A1c
Problem 8RQ
Related questions
Question
100%
Can someone help with this question?

Transcribed Image Text:Use the following table for all questions in the problem set related to glycolysis:
TABLE 18.1 Reactions and Thermodynamics of Glycolysis
AG"
AG
Reaction
Enzyme
(k/mol)
at 25°c
(kJ/mol)
Hexokinase
Glucokinase
a-D-Glucose + ATPt-=
-16.7
850
-33.9*
glucose-6-phosphate?- + ADP- + H*
Glucose-6 phosphate²-=fructose-6-phosphate-
Fructose-6-phosphate- + ATP-=
fructose-1,6-bisphosphate- + ADP- + H*
Fructose-1,6-bisphosphate
dihydroxyacetone-P- + glyceraldehyde-3-P:-
Dihydroxyacetone-P=glyceraldehyde-3-P-
Glyceraldehyde-3-P- + P?- + NAD*=
1,3-bisphosphoglycerate- + NADH + H*
1,3-Bisphosphoglycerate + ADP-
Phosphoglucoisomerase
Phosphofructokinase
+1.67
0.51
-2.92
-14.2
310
-18.8
Fructose bisphosphate
aldolase
+23.9
6.43 X 10-3
-0.23
Triose phosphate isomerase
Glyceraldehyde-3-P
dehydrogenase
Phosphoglycerate kinase
+7.56
0.0472
+2.41
+6.30
0.0786
-1.29
-18.9
2060
+0.1
3-Pglycerate- + ATP-
3-Phosphoglycerate =2phosphoglycerate
2-Phosphoglycerate-=
Phosphoglycerate mutase
+4.4
0.169
+0.83
Enolase
+1.8
0.483
+1.1
phosphoenolpyruvate-+ H,0
Phosphoenolpyruvate- + ADP- + H* =
Pyruvate + ATpt-
Pyruvate + NADH + H*= lactate" + NAD*
Pyruvate kinase
-31.7
3.63 x 10
-23.0
Lactate dehydrogenase
-25.2
2.63 x 10
-14.8
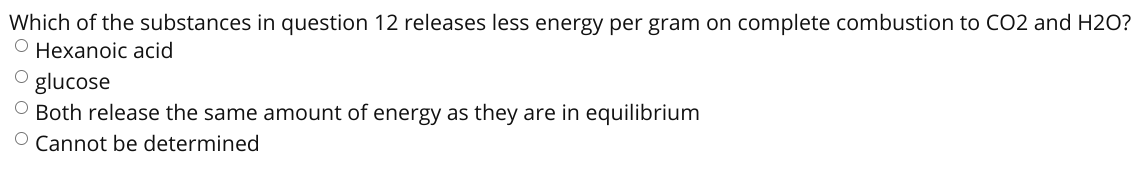
Transcribed Image Text:Which of the substances in question 12 releases less energy per gram on complete combustion to CO2 and H2O?
O Hexanoic acid
glucose
O Both release the same amount of energy as they are in equilibrium
O Cannot be determined
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 6 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

