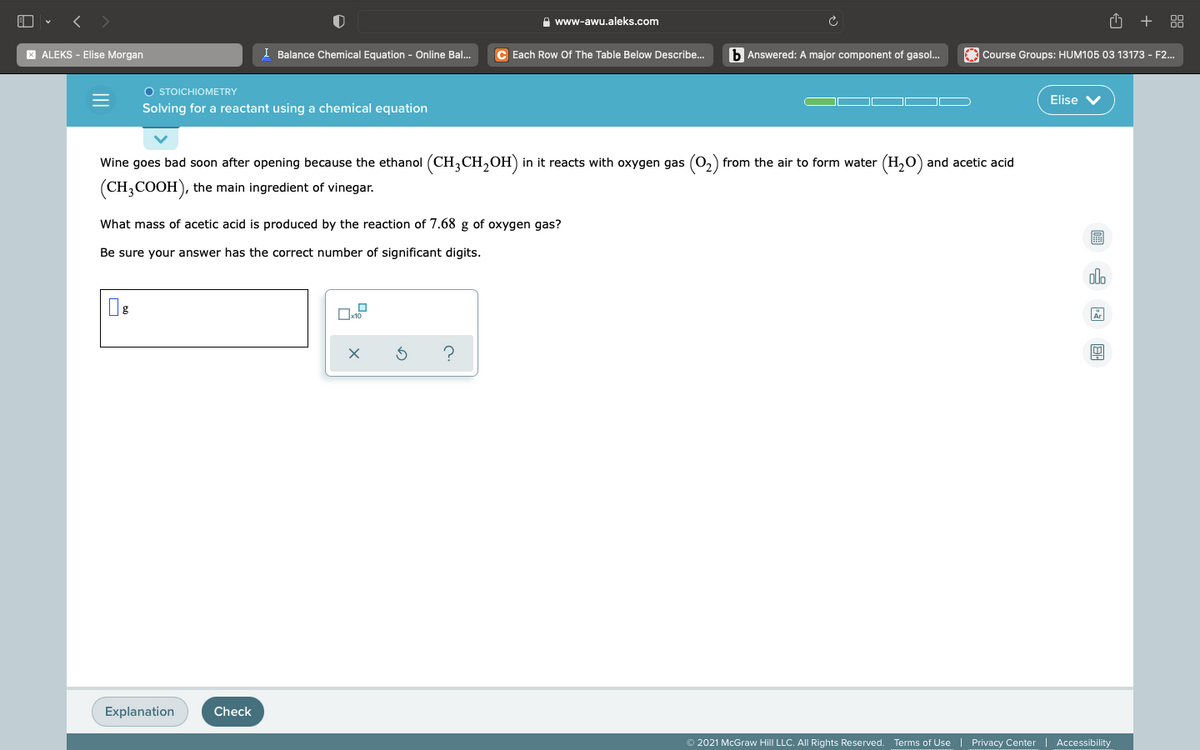
Wine goes bad soon after opening because the ethanol (CH;CH,OH) in it reacts with oxygen gas (0,) from the air to form water (H,O) and acetic acid (CH;COOH), the main ingredient of vinegar. What mass of acetic acid is produced by the reaction of 7.68 g of oxygen gas? Be sure your answer has the correct number of significant digits.
Wine goes bad soon after opening because the ethanol (CH;CH,OH) in it reacts with oxygen gas (0,) from the air to form water (H,O) and acetic acid (CH;COOH), the main ingredient of vinegar. What mass of acetic acid is produced by the reaction of 7.68 g of oxygen gas? Be sure your answer has the correct number of significant digits.
Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.14QAP
Related questions
Question
Transcribed Image Text:< >
O www-awu.aleks.com
O + 88
X ALEKS - Elise Morgan
I Balance Chemical Equation - Online Bal...
C Each Row Of The Table Below Describe..
b Answered: A major component of gasol...
Course Groups: HUM105 03 13173 - F2...
O STOICHIOMETRY
Elise v
Solving for a reactant using a chemical equation
Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,) from the air to form water (H,0) and acetic acid
(CH;COOH), the main ingredient of vinegar.
What mass of acetic acid is produced by the reaction of 7.68 g of oxygen gas?
Be sure your answer has the correct number of significant digits.
alo
Explanation
Check
O 2021 McGraw Hll LLC. All Rights Reserved. Terms of Use | Privacy Center
Accessibility
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning



Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning