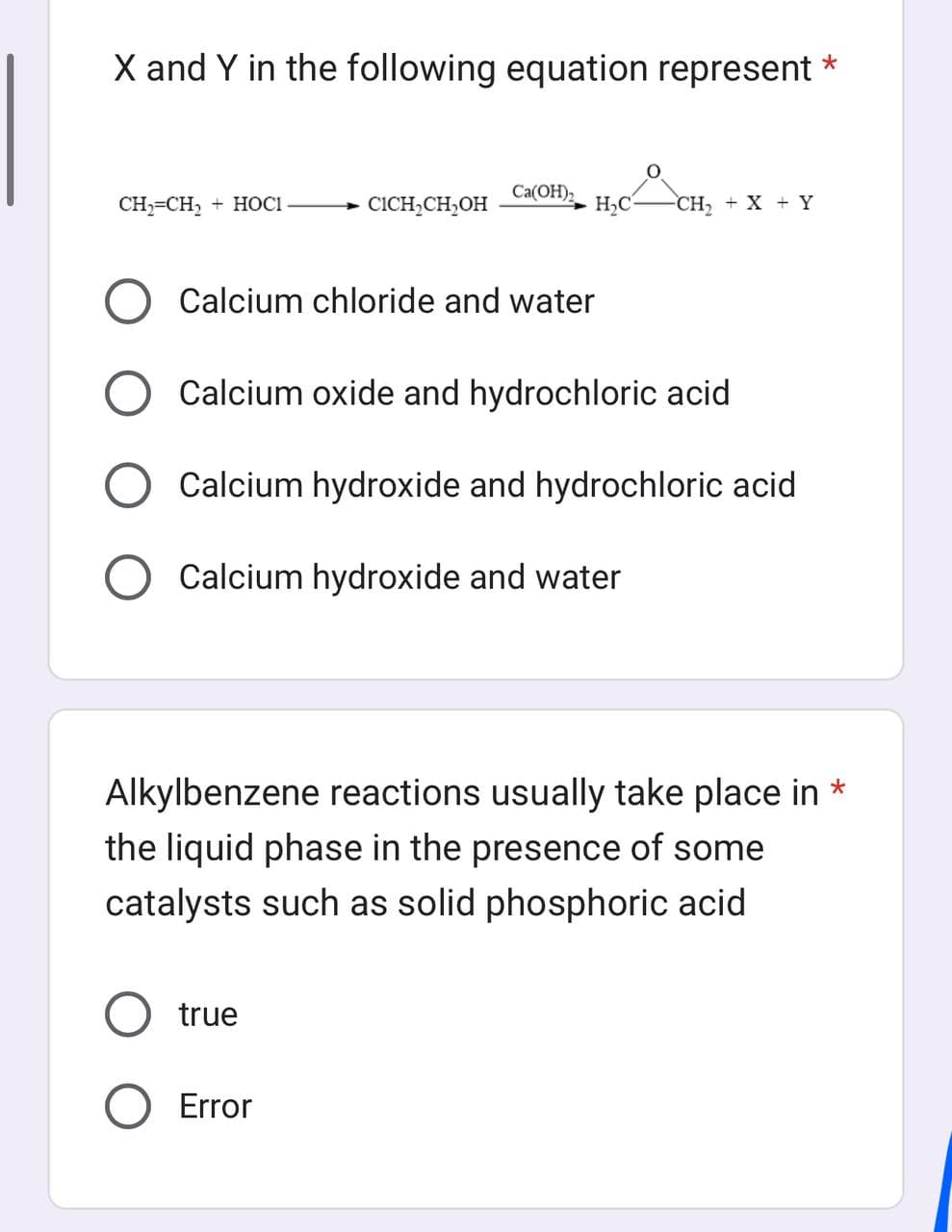
X and Y in the following equation represent * CH₂=CH₂ + HOCI CICH₂CH₂OH Ca(OH)₂ H₂C O Calcium chloride and water -CH₂ + X + Y Calcium oxide and hydrochloric acid Calcium hydroxide and hydrochloric acid O Calcium hydroxide and water
Q: 20. What is the product obtained from the following reaction? Circle one answer. OH 4 heat 3 OH 2 OH
A:
Q: 2. Propose three types of procedural error that may impact the results that were experimentally…
A: Answer. Errors generally occur while conducting experiments which can effect our results but with…
Q: Q1 / true or false 13- The wavelength of hexatriene is less than that of 1,3-butadiene in…
A: When a double-bonded molecule such as ethene (common name ethylene) absorbs light, it undergoes a π…
Q: Answer chooices are: a) +4.9 x 101 kJ b) +2.3 x 102 kJ c) -9.7 x 101 kJ d) -1.1 x 102 kJ e)…
A: Given, 3 I2 s + 2 Fe s → 2 Fe3+ aq + 6 I- aqRequired, The G of…
Q: 15. (The following problem is an actual incident) A state pharmacy inspector came to a pharmacy to…
A: Let's consider 500 mg erythromycin (MW = 733.93 g/mol) The actual formulation is 500 mg erythromycin…
Q: 10- The strongest absorption area of the benzene ring in visible and ultraviolet spectroscopy at 255…
A: A question based on spectroscopy that is to be accomplished.
Q: a. H CI @ H b. H في C. مله
A: Here we have to identify the acidic hydrogen of above compounds and write the conjugate base of that…
Q: 5) ممهدة ONO 2) CUCN A Br B N C NH₂ D
A: Organic reactions are those in which organic reactant react to form organic products.
Q: The property that describes the gradual mixing of two gases is_ cohesion effusion surface tension O…
A:
Q: The equilibrium constant, Kc, for the following reaction is 77.5 at 600 K. CO(g) + Cl₂(g) =cocl₂(g)…
A: Given- Kc = 77.5 Temperature = 600K Moles of CO = 0.351 moles Moles of…
Q: A sample of methane of mass 45.0 g occupies a container at 310 K. Calculate the work done when the…
A:
Q: Determine which of the following procedures are steps in drawing the resonance structures of…
A: For The procedural steps in drawing the resonance structures of pyridine and pyrazine Leave out…
Q: 5. Write forward and reverse reactions for the following: CH4 (g) + 2H2S (g) (g) ==== CS2 (g) + 4H2…
A:
Q: What is the name of the following alkane? A) 2,2-dimethylheptane B) 3-dimethylhexane C…
A:
Q: S O NaBH4 OLIAIH4 OBH3, THF OLIAIH(O'Bu) 3 OH
A: Organic reactions are those in which organic reactant react to form organic products.
Q: The strongest interparticle attractions exist between particles of a between particles of a O solid,…
A:
Q: 2) Show how you would accomplish the following synthetic conversions: or a. Y- b. HO S- -2 0-6 NH₂
A: Given,
Q: Which of the following is FALSE? A and B A. The half-life of a first-order reaction is…
A: The statement C is false.
Q: Solutions of lead nitrate and potassium iodide are mixed together. Calculate the value of Q for the…
A: Lead nitrate, Pb(NO3)2(aq) is completely dissociated in water to give Pb2+(aq) and 2 NO3-(aq). Hence…
Q: A compound gives the following ¹H NMR spectrum. The signal area ratio (integration) for the three…
A: Two questions based on NMR spectroscopy that are to be accomplished.
Q: 14) The intensity of a signal in a ¹H NMR spectrum is determined by A. The number of neighboring C.…
A: In the given question we have to select the correct option from the following. the intensity of…
Q: Consider the reaction: CO(g) + H₂O(l) → CO₂(g) + H₂(9) - Using standard thermodynamic data at 298 K,…
A:
Q: Aluminium is reacted with hydrochloric acid to produce hydrogen gas and an aqueous solution of…
A: Given, Aluminium (Al) is reacted with hydrochloric acid (HCl) to produce hydrogen gas (H2) and an…
Q: Large amounts of titanium metal are refined by reacting titanium(III) chloride solution with…
A: Given mass of Mg=1.46kg Given mass of Ti=1.62kg
Q: The very high melting point and boiling point of ionic compounds such as NaCl is due to the very…
A: • We need to determine which intermolecular force of attraction exist in ionic compounds.
Q: Solve for x, where M is molar and s is seconds. x = = (6.1 × 10³ M-²s¯¹)(0.25 M)³ Enter the answer.…
A: Please find the attachment.
Q: What is the major product of this reaction? O H HCN ???
A:
Q: At 20 °C a saturated solution of silver acetate, AgCH, CO2, contains 1.0 g of the silver compound…
A:
Q: Propose routes to the following compounds from propyl bromide and any necessary organic or inorganic…
A:
Q: Complete and balance the following half-reaction in acidic solution. Be sure to include the proper…
A:
Q: Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic.…
A: Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic…
Q: The coefficient of fugacity of percent gas at 290 K and 2.1 MPa is 0.68. Calculate the molar Gibbs…
A:
Q: Which is the correct half reaction for the oxidation of S₂03² to S406²?
A:
Q: Indicate the acid and its conjugate base, and the base and its conjugate acid. HC1O4 (aq) + H₂O(l) →…
A: Since, Base on addition of H+ formed conjugate acid. Acid on removal of H+ formed conjugate base.…
Q: Use the References to access important values if needed The hydroxide ion concentration in an…
A: #Q.1: Given the hydroxide ion concentration, [OH-(aq)] = 0.054 M
Q: 15) The positively polarized carbon atom of a carbonyl group acts as: A) an electrophile and a Lewis…
A: we have to identify the correct statement for the carbon atom of carbonyl group
Q: In the following molecule, which bond(s) is (are) a peptide bond? Mark all that apply. DA ОВ BC O D…
A: The peptid bond is an amide type bond which is formed by the condensation of two amino acids through…
Q: a) What terms can arise from the (He)2s22p13d1 excited configuration of carbon? Terms: b) What…
A:
Q: An organic acid has pka = 2.87. What is its Ką value?
A:
Q: Intensity (a.u.) 4000 3352 cm¹ 3500 3000 UNKNOWN B 1710 cm¹ 1668 c cm¹ 2500 Wavenumber (cm¹¹) 2000…
A: Ir spectroscopy helps in the identifiaction of organic compounds.It helps in determining functional…
Q: Can you explain the mechanism? Can you provide a shortcut?
A:
Q: What is the name of the intermolecular interaction between two charged functional groups (one…
A: Charged atoms or molecules are called ions. Two charged molecules interact with each other via…
Q: What is the molar solubility of mercury (I) fluoride? Ksp = 3.1 x 10-6.
A:
Q: Which is the net ionic equation for the oxidation of S₂O32- to S4062- and the reduction of iodine to…
A: we have to write the net ionic equation for the given oxidation and reduction reaction
Q: Ethanol (C₂H6O) burns in air C₂H6O(1) +O2(g) → CO2(g) + H₂O (1) Balance the equation and determine…
A:
Q: Find the standard thermodynamic values Enthalpy, Entropy and the free energy of the zinc-carbon…
A: Enthalpy ,Entrophy and gibbs free energy are related by following equation: ∆G° =∆H° -T∆S°where ∆H°…
Q: A reaction has an activation energy of 225,0 kJ/mol. By what factor will the rate constant, k,…
A: Arrhenius equation: Arrhenius equation give the relationship between the activation energy and the…
Q: 8) When pyridine is treated with a mixture of nitric and sulfuric acids, the major EAS product is:…
A:
Q: The 0-16 nucleus has a mass of 15.9905 amu. A proton has a mass of 1.00728 amu, a neutron has a mass…
A: Given the mass of O-16 nucleus = 15.9905 amu mass of a proton = 1.00728 amu mass of a neutron =…
Q: 13-when changing the polarityof solvent frome non-polar .the value of the transition wavelength…
A: In the given question we have to choose the correct option from the following. when changing the…
Step by step
Solved in 3 steps with 1 images

- The Tris buffer system is commonly used in biochemistry as its pKa of 8.1 allows it to buffer close to physiological pH. (CH2OH)3CNH2 + H+ Û (CH2OH)3CNH3+ What concentrations of Tris and Tris H+ are obtained in a 200 mL solution at pH 8.20 containing 6.1g of Tris? The molecular mass of Tris is 121.1 gmol-1 Please do this step by step thank you!calculate the ph of a 0.465 M solUTION OF ethylamine CH3CH2NH2 and 0.211 M ethylammonium bromide CH3CH2NB3Br. the base dissociation constant kb is 6.31 x 10 -4. what is the pH if 76.0 ml of 0.150 M of a strong acid like HCI is added to 100 ml of buffer solution. what if a strong base like NaOh is added with the same ammount insteadCalculate the ratio of dissolved benzoic acid to benzoate ion that will exist in solution at equilibrium at a pH of 2.00. The acid equilibrium constant (K a ) for benzoic acid is 6.46 * 10 ^ (- 5) . Note that this calculation doesn't account for the benzoic acid that has precipitated from solution, but you can assume that the higher the ratio, the more benzoic acid will precipitate. Then, Carry out the same calculation for a pH of 4.00, and explain why it was important to reduce the pH to below 4 in this experiment.
- Hydrogen sulfide (H2S) is an odorous gas that can be stripped from solution by bubbling air through the solution. H2S when dissolved in water is also a weak acid that can deprotonate to bisulfide ions which are not removed by air stripping. That reaction is ?2?--> H^+ + ??^- With equilibrium constant Ka=0.86x10-7.Find the fraction of hydrogen sulfide in the H2S form at pH 6 and pH 8. At which pH would you expect to have an easier time to strip away H2S gas?Standard solution : 0.201 grams of acetylsalicylic acid was combined with 10 mL of 0.5 M NaOH, then transfered to a 100 mL flask and filled the rest of the way with water Then 0.500 mLs of the standard solution was transfered to a 10.00 mL flask and diluted to the 10 mL mark with 0.02 M of buffered iron chloride. Number of moles of NaOH = 0.005 moles Since the reaction between acetylsalicylic acid and NaOH is a 1:1 reaction, the number of moles of acetylsalicylic acid used in the reaction is also 0.005 moles. the concentration from analysis question 5: acetylsalicylic acid in the 100.00 mL volumetric flask is 0.050 M. From your plot, what is the value of εb? From the Beer- Lambert law For the aspirin sample, calculate the concentration of acetylsalicylic acid present using the value of εb that you found The concentration from analysis question 5 represents the concentration in the 10.00 mL sample that was prepared in the volumetric flask using an aliquot of the solution in the…You are conducting a biochemical experiment with an enzyme that has optimal activity at pH = 6.50. You decide to use carbonate (pKa1 = 6.38, pKa2 = 10.30) as the buffer to keep the pH stable throughout the enzymatic reaction. (Recall that the formula for carbonic acid is H2CO3.) You prepare a 0.4 M solution of carbonate buffer at pH = 10.50. Calculate the concentrations of the major carbonate species in your solution. Show your calculations.
- Write the equilibrium constant expressions for the following reactions and determine the value for the equilibrium constants AgCl + 2NH3(aq) ⇌ Ag(NH3)2+ +Cl-(aq) BaCO3 +2H3O+(aq) ⇌ Ba2+(aq) + H2CO3(aq) +2H2O(l) The basic dissociation of aniline, C6H5NH2 The acidic dissociation of methyl ammonium hydrochlorideAlizarin yellow R, Ka=7.9×10−12, is yellow in its protonated form (HX) and red in its ionized form (X−). At what pH will alizarin yellow R be a perfect orange color?Calculate the pH of 640. mL of a 0.280-M solution of hydrocyanic acid before and after the addition of 0.116 mol of sodium cyanide.pH befor addition = pH after addition =
- At 25oC, Ka for acid dissociation of lactic acid (HC3H5O3) is 1.0 x 10-4. Calculate ΔGo for the reaction HC3H5O3 (aq) + H2O(l) H3O+(aq) + C3H5O3-(aq)Tris(hydroxymethyl)aminomethane [(HOCH2)3CNH2—Tris, or THAM] is a weak base frequently used to prepare buffers in biochemistry. Its Kb is 1.2 × 10−6 and pKb is 5.92. The corresponding pKa is 8.08, which is near the pH of the physiological buffers, and so it exhibits good buffering capacity at physiological pH. What weight of THAM must be taken with 100 mL of 0.50 M HCl to prepare 1 L of a pH 7.40 buffer?10.0 mL of 0.100 M HCl solution was added to 20.0 mL of 0.100 M (CH3CH2)3N (triethylamine). Kb (CH3CH2)3N = 5.2 x 10–4 Determine the following: a) Concentration of (CH3CH2)3N that did not react with acid. b) Concentration of (CH3CH2)3NH+ formed from the reaction with acid. c) pOH.

