
Which of the following would you expect to be more soluble in water than in benzene? (Select all that apply.)
(a)
(b)
(e)
Interpretation:
The compounds that is more soluble in water than benzene is/are to be identified.
Concept introduction:
The various kinds of interactions that bind a molecule are known as intermolecular forces. These can be dispersion, dipole–dipole, ion–dipole,and hydrogen bonding.
The solubility of a compound depends on the intermolecular forces present. Apolar solvent is soluble in apolar solvent only. A compound is soluble in water only when it is able to form hydrogen bonds with it.
Answer to Problem 1KSP
Solution: The correct answersareoption (a) and option (e).
Explanation of Solution
Reason for the correct option:
The molecule, methanol
Thus, it is soluble in water more readily. On the other hand, benzene is a nonpolar molecule. So, methanol is insoluble in benzene.
Potassium iodide
Hence, option (a) and option (e) are correct.
Reasons for the incorrect options:
Option (b) is incorrect because
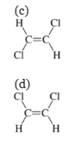
Option (c) is incorrect because trans-dichloroethene is a nonpolar molecule. It is insoluble in water. So, this option is incorrect.
Option (d) is incorrect because cis-dichloroethene is a polar molecule but it is unable to form hydrogen bonds in water. It is, therefore, insoluble in water. So, this option is incorrect.
Hence, options(c) and (d) are incorrect.
Want to see more full solutions like this?
Chapter 11 Solutions
CHEMISTRY(LOOSELEAF W/CODE)- CUSTOM
- In terms of the kinetic molecular theory, in what ways are liquids similar to gases? In what ways are liquids different from gases?arrow_forwardIn terms of the kinetic molecular theory, in what ways are liquids similar to solids? In what ways are liquids different from solids?arrow_forwardThe normal boiling point of SO2 is 263.1 K and that of NH3 is 239.7 K. At −40 °C, would you predict that ammonia has a vapor pressure greater than, less than, or equal to that of sulfur dioxide? Explain.arrow_forward
- The later halogens form pentafluorides ClF5, BrF5 and IF5 at 0oC, one of these is a solid, one a liquid and one a gas. specify which is which and explain how you reached the conclusion.arrow_forwardA 0.0896g of H2O us placed in a 250ml sealed container. Is there any liquid present when temperature is 70 celsius. I got yes, cuz my answer is 0.56atm which is higher than the vapor pressure of water that is 0.308. However, how do I calculate the mass of water being condensed?arrow_forwardWhich of the following compounds would need to overcome dispersion forces when converting from a liquid to a gas? Select all that apply. PH3, I2, SO2, CO2 . Which of the following compounds would be expected to form intermolecular hydrogen bonds in the liquid state? Select all that apply. NH3, H2Se, CH3OH, CH3Clarrow_forward
- Which compound would you expect to have greater surface tension: acetone [(CH3)2CO] or water (H2O)? Explain how?arrow_forwardWhich compound has a higher boiling point: butane, C4 H10, or hexane, C6H4?arrow_forward2. A closed vessel of volume 10.0 L initially contains 1.303 g of water and no water vapor. Please calculate the mass of liquid water remaining once the system reaches equilibrium at 30 degrees Celsius. The vapor pressure of water at that temperature is equal to 0.0418 atm.arrow_forward
- As the temperature increases, do you expect the surface tension ofa liquid to increase, decrease, or stay the same?arrow_forwarddoes LiF have a higher vapor pressure at 25oC than H2S?arrow_forwardacetone has Pvap = 40mmHg at -9.4C and a normal boiling point of 56.5C. what is deltaHvap for acetone in kJ/mol?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





