
General, Organic, & Biological Chemistry
3rd Edition
ISBN: 9780073511245
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 11.6, Problem 11.20P
Indicate the polar bonds in each compound. Label the bonds with
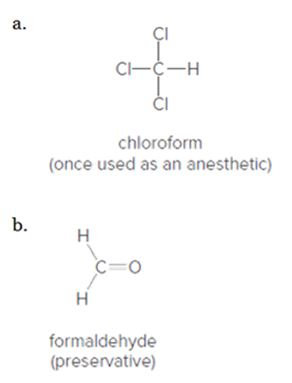
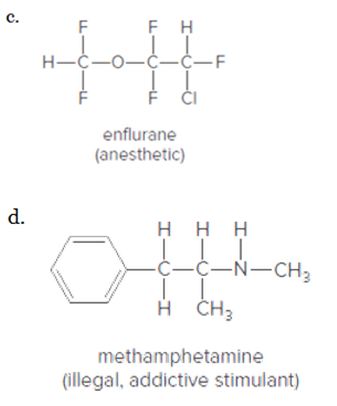
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Classify the following bonds as nonpolar, polar, or ionic: (a) the bond in ClF, (b) the bond in CsBr, and (c) the carbon-carbon double bond in C2H4.
Which molecule contains polar covalent bonds and non-polar covalent bonds?
Na2S
Cl2
CCl4
CH3OH
CH3CH3
Rank these bonds in order of decreasing polarity: Al-Al C-O C-H Si-O Li-O
Chapter 11 Solutions
General, Organic, & Biological Chemistry
Ch. 11.1 - Prob. 11.1PCh. 11.2 - Fill in all H's and lone pairs in each compound.Ch. 11.3 - Prob. 11.3PCh. 11.3 - Prob. 11.4PCh. 11.3 - Prob. 11.5PCh. 11.3 - Prob. 11.6PCh. 11.3 - How many lone pairs are present in lidocaine, the...Ch. 11.4 - Convert each compound to a condensed formula.Ch. 11.4 - Convert each condensed formula to a complete...Ch. 11.4 - Convert each skeletal structure to a complete...
Ch. 11.4 - How many H’s are bonded to each indicated carbon...Ch. 11.5 - Prob. 11.12PCh. 11.5 - For each compound: [1] Identify the functional...Ch. 11.5 - Prob. 11.14PCh. 11.5 - Prob. 11.15PCh. 11.5 - Prob. 11.16PCh. 11.5 - Identify all of the functional groups in atenolol,...Ch. 11.5 - Prob. 11.18PCh. 11.5 - Prob. 11.19PCh. 11.6 - Indicate the polar bonds in each compound. Label...Ch. 11.6 - Prob. 11.21PCh. 11.6 - Prob. 11.22PCh. 11.6 - Predict the water solubility of each compound....Ch. 11.6 - Prob. 11.24PCh. 11.7 - Prob. 11.25PCh. 11.7 - Prob. 11.26PCh. 11 - Prob. 11.27PCh. 11 - Prob. 11.28PCh. 11 - Complete each structure by filling in all H’s and...Ch. 11 - Prob. 11.30PCh. 11 - Prob. 11.31PCh. 11 - Prob. 11.32PCh. 11 - Prob. 11.33PCh. 11 - Prob. 11.34PCh. 11 - “Ecstasy” is a widely used illegal stimulant....Ch. 11 - Prob. 11.36PCh. 11 - Explain why each C—C—C bond angle in benzene...Ch. 11 - Prob. 11.38PCh. 11 - Prob. 11.39PCh. 11 - Prob. 11.40PCh. 11 - Convert each compound to a skeletal structure.Ch. 11 - Convert each compound to a skeletal structure.Ch. 11 - Convert each shorthand structure to a complete...Ch. 11 - Convert each shorthand structure to a complete...Ch. 11 - A and B are ball-and-stick models of two compounds...Ch. 11 - Prob. 11.46PCh. 11 - What is wrong in each of the following shorthand...Ch. 11 - Prob. 11.48PCh. 11 - Prob. 11.49PCh. 11 - Albuterol (trade names Proventil and Ventolin) is...Ch. 11 - Prob. 11.51PCh. 11 - Prob. 11.52PCh. 11 - Prob. 11.53PCh. 11 - Prob. 11.54PCh. 11 - Prob. 11.55PCh. 11 - GHB is an addictive, illegal recreational drug...Ch. 11 - Prob. 11.57PCh. 11 - Prob. 11.58PCh. 11 - Prob. 11.59PCh. 11 - Prob. 11.60PCh. 11 - Prob. 11.61PCh. 11 - Prob. 11.62PCh. 11 - You are given two unlabeled bottles of solids, one...Ch. 11 - State how potassium iodide (KI) and pentane...Ch. 11 - The given beaker contains 100 mL of the organic...Ch. 11 - Prob. 11.66PCh. 11 - Why do we need to know the shape of a molecule...Ch. 11 - 1,1-Dichloroethylene (CH2=CCl2) is a starting...Ch. 11 - Indicate the polar bonds in each molecule. Label...Ch. 11 - Indicate the polar bonds in each molecule. Label...Ch. 11 - Classify each molecule as polar or nonpolar.Ch. 11 - Classify each molecule as polar or nonpolar. a....Ch. 11 - Which molecule is more water soluble? Explain.Ch. 11 - Explain why pantothenic acid, vitamin B5, is water...Ch. 11 - Prob. 11.75PCh. 11 - Prob. 11.76PCh. 11 - Explain why regularly taking a large excess of a...Ch. 11 - You can obtain the minimum daily requirement of...Ch. 11 - Prob. 11.79PCh. 11 - Vitamin B6 is obtained by eating a diet that...Ch. 11 - Prob. 11.81PCh. 11 - Can an oxygen-containing organic compound, have...Ch. 11 - Prob. 11.83PCh. 11 - Prob. 11.84PCh. 11 - Benzocaine is the active ingredient in topical...Ch. 11 - Methyl salicylate is responsible for the...Ch. 11 - Answer the following questions about aldosterone,...Ch. 11 - Answer the following questions about...Ch. 11 - Prob. 11.89PCh. 11 - Skin moisturizers come in two types, (a) One type...Ch. 11 - THC is the active component in marijuana (Section...Ch. 11 - Cocaine is a widely abused, addicting drug....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the following bonds from most polar to least polar?Cl-O, Mg-O, O-O, C-Oarrow_forwardBased on their positions in the periodic table, which of the following bonds is the most polar? O-F C-F B-F F-Farrow_forwardUse the symbols d+ and d- to show the direction of the polarity of the indicated bond in each of thefollowing compounds F-Br H2N-OH H3C-NH2arrow_forward
- Which type of bond is most likely to form between two atoms of Carbon? A. Ionic B. Polar Covalent C. Nonpolar Covalent D. Hydrogenarrow_forwardWhich molecule or compound below contains a pure covalent bond? PF3 NaCl Cl2 Li2CO3 SCl6arrow_forwardWhich structure best describes the polarity of an H-O bond?arrow_forward
- Which of the following molecules has/ have resonance structures? Select one or more: a.CH2=CH2 b.CH3–OH c.CH2=CH–O– d.CH3–O–CH3arrow_forwardWhich compounds contain polar covalent bonds?arrow_forwardConsidering the position of the elements in the periodic table and their relative electronegativities and bond polarities, which bond is longest? a. carbon - Oxygen triple bond b. carbon - Oxygen single bond c. carbon - Carbon single bond d. carbon - Carbon double bond e. carbon - Nitrogen triple bond Which bond is the strongest? a. carbon - Nitrogen triple bond b. carbon - Nitrogen double bond c. carbon - Hydrogen bond d. carbon - Carbon triple bond e. carbon - Carbon single bondarrow_forward
- Identify any polar covalent bonds by drawing dipole arrows in the appropriate locations.arrow_forward1.) Complete the following properties of methyl salicylate: a. Polarity (polar/non-polar) b. Bond lengthsarrow_forwardIs C3H7Br a polar molecule? If so, indicate the polarity of each bond and the direction of the net dipole.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Types of bonds; Author: Edspira;https://www.youtube.com/watch?v=Jj0V01Arebk;License: Standard YouTube License, CC-BY