
Concept explainers
- The numbers of orbiting electrons in aluminum and silver are 13 and 47, respectively. Draw the electronic configuration for each, and discuss briefly why each is a good conductor.
- Using the Internet, find the atomic structure of gold and explain why it is an excellent conductor of electricity.
(a)
To draw:
The electronic configuration of aluminum and silver. Also, state the reason due to which both aluminum and copper are considered a good conductor.
Explanation of Solution
Given:
The numbers of orbiting electrons in aluminum and silver are
The arrangement of electron inside the atom and outside the nucleus follows a particular pattern. There is a shell structure in which all electron falls and the energy of the electrons decide which shell to fall. The number of electrons in each shell is always
In shell
In Shell
Inside shell, actually electron falls in sub-shell. The name of sub-shell are
In a nutshell, the larger the value of
The electronic configuration of any element is based on below pattern:
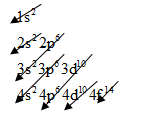
For aluminum, we know that the numbers of orbital electrons are 13.
So, arrange the electrons will be as below
The electronic configuration of Aluminum is
Here we can see, Aluminum has one free electron in its valence shell. Therefore, this free electron can move easily and hence, this causes the higher conductivity for Aluminum.
For silver, we know that the numbers of orbital electrons are 47.
So, arrange the electrons will be as follows:
The electronic configuration of silver is
Silver is a good conductor because it has free electrons in its valence shell. It is clearly seen from the electronic configuration of silver (
(b)
The atomic structure of the gold. Also, state the reason due is an excellent conductor of electricity.
Explanation of Solution
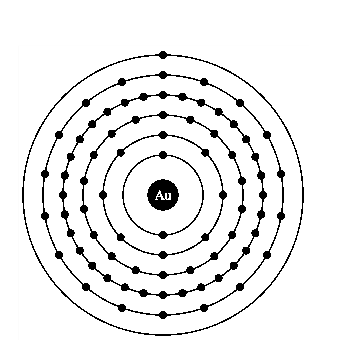
Atomic structure of gold.
The electronic configuration of any element is based on below pattern:
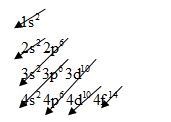
For gold, we know that the numbers of orbital electrons are
So, arrange the electrons for gold will be as follows:
The electronic configuration of gold is
Gold is a good conductor of electricity because it has free electrons in its valence shell.
It is clearly seen from the electronic configuration of gold that, s orbit is holding only one electron and maximum limit is up to
Want to see more full solutions like this?
Chapter 2 Solutions
Introductory Circuit Analysis
Additional Engineering Textbook Solutions
Fundamentals of Applied Electromagnetics (7th Edition)
Electrical Engineering: Principles & Applications (7th Edition)
C How to Program (8th Edition)
Programming in C
Introduction to Java Programming and Data Structures, Comprehensive Version (11th Edition)
Web Development and Design Foundations with HTML5 (8th Edition)
- Determine the absolute potential in air at a distance of 3cm from a point charge of 500 uC. *arrow_forwardconducting line on an IC chip is (2.8 mm) long, and has a rectangular cross-section (1x 4 um). A current of (5 mA) produces a voltage drop of (100 mV) across the line. Determine the electron concentration given that the electron mobility is (500 cm²/V.Sec.).arrow_forwardHow to compute the atomic packing factor for the diamond cubic structure? detailed example with solution pleasearrow_forward
- Find the distance of an isolated positive point charge of 10 nC, in order for it to produce an electric potential of 120 V.arrow_forwardThe electron number density in a semiconductor varies from 1020 m-3 to 1012 m-3 linearly over a distance of 4 μ Determine the electron diffusion current and electric field at the midpoint if no current flows, μе = 0.135 m2V-1s-1 and T = 300 K.arrow_forwardA copper wire has a diameter of 2.05 mm and carries a current of 15 A due solely toelectrons. (These values are common in residential wiring.) Each electron has a charge of -1.60 x 10-19 C. Assume that the free-electron (these are the electrons capable of movingthrough the copper) concentration in copper is 1029 electrons/m³. Find the average velocity ofthe electrons in the wire.arrow_forward
- (a) Compute the electrical conductivity of a 5.1mm diametercylindrical silicon specimen 51 mm long in which a current of0.1 A passes in an axial direction. A voltage of 12.5 V ismeasured across two probes that are separated by 38 mm.(b) Compute the resistance over the entire 51 mm of thespecimen.arrow_forwardProblem 10. Given a piece of Si doped with B to a level of 1015 atoms/cm3, (a) what carrier moves with a higher mobility (including why), and (b) How would this change if the dopant were P instead of B?arrow_forwardA cylindrical copper wire transmits a charge of 360 °C in 10 minutes. Copper contains 8.5x10^28 free electrons per cubic meter. If the radius of the wire is 1mm, what is the drift velocity of the electrons (in mm/s)? (take e=1.6x10-19C, pi=3!)arrow_forward
- 5. A long copper wire carries a current of 20A which is evenly distributed in each cross-section of the wire. If the diameter of the wire is 4 mm, and the resistance per unit length is 5 Q2/km, calculate the energy density of the magnetic and electric fields on the surface of the wire!arrow_forwardSilicon is doped with ND= 2 × 1015 impurity atoms/cm3 . Assume the electron and hole mobilities for the given impurity are un = 1320 cm2 / V.s, and up=460 cm2 /V.s respectively. Use ni = 1.5 × 1010/cm3 Calculate the resistivity of silicon. Assume an aluminum line runs diagonally from one corner of a 20 mm × 20 mm silicon material to the other corner. What is the resistance of this line if it is 1 μm thick and 5 μm wide? The resistivity of pure aluminum is 2.82 μΩ-cm.arrow_forwardIn a n-type semiconductor, the electron concentration increases linearly along the x-axis from 10^16 cm^-3 at x=0 (point A) to 5x10^20 cm^-3 at x=0.5cm (point B). In the absence of any external applied electric field, estimate the approximate diffusion current density and its direction (A to B or B to A). Assume that the electron diffusion constant, Dn = 10cm^2/s.arrow_forward
 Electricity for Refrigeration, Heating, and Air C...Mechanical EngineeringISBN:9781337399128Author:Russell E. SmithPublisher:Cengage Learning
Electricity for Refrigeration, Heating, and Air C...Mechanical EngineeringISBN:9781337399128Author:Russell E. SmithPublisher:Cengage Learning
