
Concept explainers
Where on the following outline of a periodic table do the indicated elements or groups of elements appear?
(a) Alkali metals
(b) Halogens
(c) Alkaline earth metals
(d)
(e) Hydrogen
(f) Helium
(g) Metalloids
Interpretation:
The given elements or group of elements occurs where in the periodic table has to be given.
Explanation of Solution
The elements and groups are indicated below,
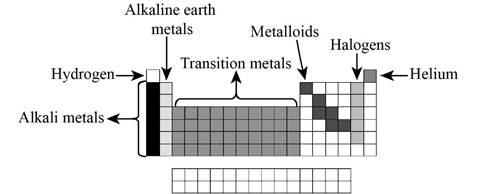
Alkali metals are 1A group elements (other than hydrogen), Halogens are 7A group elements, Alkaline earth metals are 2A group elements, Transition metals are 1B to 8B group elements, Hydrogen is the first element in the periodic table, helium is the second element in the periodic table located in 8A group. Metalloids are main group elements (which is located in 3A to 8A).
Want to see more full solutions like this?
Chapter 2 Solutions
EBK FUND.OF GENERAL,ORG.+BIO.CHEMISTRY
- How many electrons must a nitrogen atom, Z = 7, gain to attain a noble gas configuration?arrow_forwardWhat subatomic particle do all Carbon atoms, isotopes and ions have in common?arrow_forwardAnother major use of bismuth has been as an ingredient in low-melting metal alloys, such as those used in firesprinkler systems and in typesetting. The element itself is a brittle white crystalline solid. How do these characteristicsfit with the fact that bismuth is in the same periodic group with such nonmetallic elements as nitrogenand phosphorus?arrow_forward
- why do the sizes of the transition elements change more gradually than those of the representative elements?arrow_forwardPolonium is a rare element with 33 radioisotopes. The most common one, 210Po, has 82 protons and 128 neutrons. When 210Po decays, it emits an alpha particle, which is a helium nucleus (2 protons and 2 neutrons). 210Po decay is tricky to detect because alpha particles do not carry very much energy compared to other forms of radiation. For example, they can be stopped by a single sheet of paper or a few inches of air. That is one reason that authorities failed to discover toxic amounts of 210Po in the body of former KGB agent Alexander Litvinenko until after he died suddenly and mysteriously in 2006. What element does an atom of 210Po become after it emits an alpha particle?arrow_forwardElements have varying numbers of protons, neutrons, and electrons.True or false?arrow_forward

 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning


