
Concept explainers
The fatty acid composition of three triacylglycerols (A, B, and C) is reported below. Predict which one has the highest melting point. Which one do you expect to be liquid (oil) at room temperature? Explain.
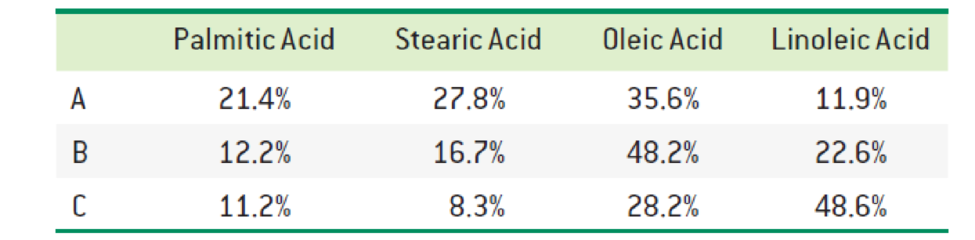
Interpretation:
The triacylglycerols with highest melting point and fatty acid expected to be liquid at room temperature has to be identified.
Concept Introduction:
Triacylglycerol:
It is also known as triglyceride, an ester composed of glycerol and long chains of acyl fatty acids.
Explanation of Solution
The fatty acid composition A has the highest melting point and B and C are expected to be at low melting point.
Since the composition A contains
The triacylglycerols with highest melting point and fatty acid expected to be liquid at room temperature was identified.
Want to see more full solutions like this?
Chapter 23 Solutions
Modified Masteringchemistry With Pearson Etext -- Valuepack Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry
- 70 degree Celcious is the melting point of stearic acid, and 4 degree Celcius is the melting point of oleic acid. Explain using the concept of lipids why their melting point is so different.arrow_forwardDefine the following terms: a. monounsaturated fatty acid b. polyunsaturated fatty acid c. saturated fatty acid d. nonessential fatty acid e. essential fatty acidarrow_forwardIn a dietary context, what is the difference between good and bad cholesterol?arrow_forward
- Why do unsaturated fatty acids have lower melting points than their saturated counterparts? What is relationship of the degree of unsaturation and melting or boiling point?arrow_forwardWhich of the following statements concerning glycolipids is true? a. All glycolipids contain sphingosine. b. All glycolipids contain one sugar residue. c. All glycolipids contain phosphatidylglycerol. d. Some glycolipids contain ceramide, while others do not.arrow_forwardWhy are polyunsaturated omega 3 acids considered as good for our health? Explain such phenomena based on their chemical structurearrow_forward
- Draw an example of an unsaturated fatty acid in both the cis and trans configuration. Number the carbons using the wconvention. Indicate which will have the higher melting pointarrow_forwardIn what chemical form does cholesterol usually exist in the bloodstream?arrow_forwarda.) identify each of the following as a saturated, monounsaturated, polyunsaturated, omega-3, or omega-6 fatty acid b.) write the shorthand notations for all the fatty acidsarrow_forward
- Describe the differences between saturated fatty acids and unsaturated fatty acids in terms of their chemical structures. What is a polyunsaturated fatty acidarrow_forwardWrite an equation for lipids with KOH and heat and then explain any differences between the three classes of lipids (phospholipids, triacylglycerol and cholesteral) on heating the lipids with KOH and then shaking while referring to the equationarrow_forwardGive the lipid number for the following fatty acid:arrow_forward
