
Concept explainers
For alanine,
(a) 2.00.
(b) 6.00.
(c) 10.50.
What is the principal species at each pH?
(a)
Interpretation:
The value of
Concept introduction:
General formula of alanine is shown as follows:
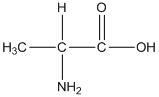
The
Answer to Problem 29QAP
The principal species at
Explanation of Solution
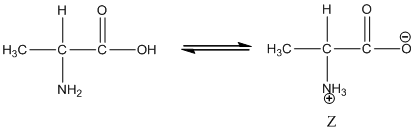
Under equilibrium condition, the Zwitter ionic form of alanine is shown as follows:
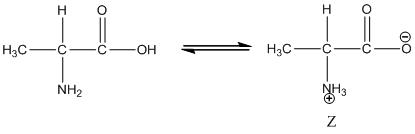
Under acidic condition, oxygen atom accepts the proton Zwitter ion exists in cationic form.
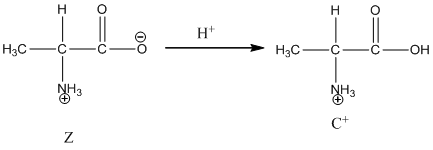
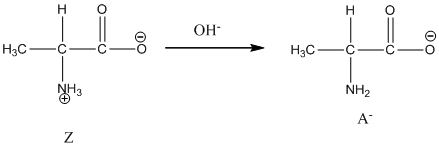
Under basic condition, base abstracts proton form nitrogen atom and it forms the anionic form.
Dissociation of cation is represented as follows:
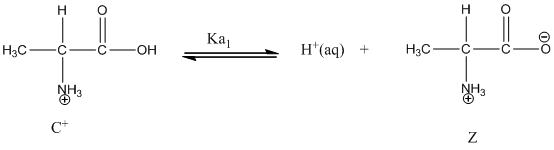
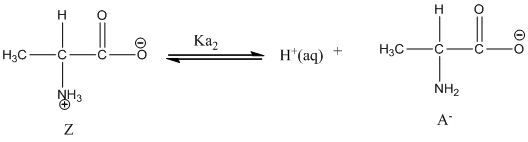
Dissociation of anion is represented as follows:
Given value of
At
So,
Take antilog both sides,
Here, Z is Zwitter ion and C is cationic form of Zwitter ion.
Here, Z is Zwitter ion and A is anionic form of Zwitter ion.
Therefore,
The principal species at
(b)
Interpretation:
The value of
Concept introduction:
General formula of alanine is shown as follows:
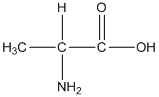
The
Answer to Problem 29QAP
The principal species at
Explanation of Solution
Under equilibrium condition zwitter ionic form of alanine is:
Under acidic condition oxygen atom accepts the proton zwitter ion exists in cationic form.
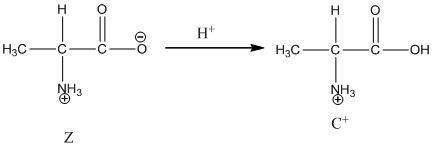
Under basic condition base abstracts proton form nitrogen atom and it forms the anionic form.
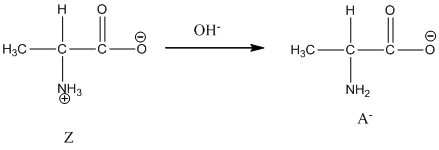
Dissociation of cation is-
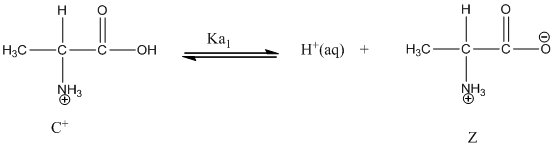
Dissociation of anion is-
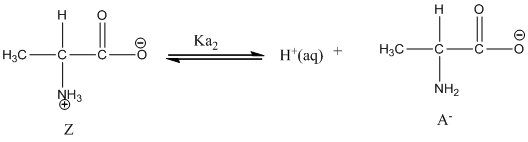
Given value of
At
So,
Take antilog both sides,
Here, Z is zwitter ion and C is cationic form of zwitter ion.
Here, Z is zwitter ion and A is anionic form of zwitter ion.
The principal species at
(c)
Interpretation:
The value of
Concept introduction:
General formula of alanine is shown as follows:
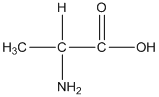
The
Answer to Problem 29QAP
The principal species at
Explanation of Solution
Under equilibrium condition zwitter ionic form of alanine is:
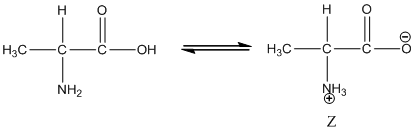
Under acidic condition oxygen atom accepts the proton zwitter ion exists in cationic form.
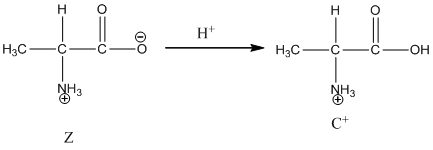
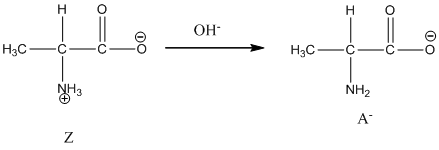
Under basic condition base abstracts proton form nitrogen atom and it forms the anionic form.
Dissociation of cation is-
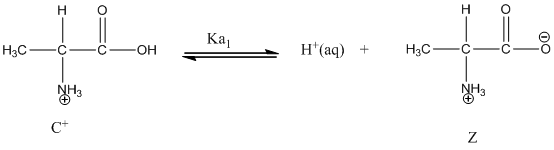
Dissociation of anion is-
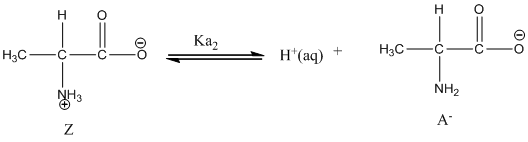
Given value of
At
So,
Take antilog both sides,
Here, Z is zwitter ion and C is cationic form of zwitter ion.
Here, Z is zwitter ion and A is anionic form of zwitter ion.
The principal species at
Want to see more full solutions like this?
- 22-59 What is the effect of salt bridges on the tertiary structure of proteins?arrow_forward22-49 Based on your knowledge of the chemical properties of amino acid side chains, suggest a substitution for leucine in the primary structure of a protein that would probably not change the character of the protein very much.arrow_forward22-89 What kind of noncovalent interaction occurs between the following amino acids? (a) Valine and isoleucine (b) Glutamic acid and lysine (c) Tyrosine and threonine (d) Alanine and alaninearrow_forward
- 22-20 Show how alanine, in solution at its isoelectric point, acts as a buffer (write equations to show why the pH does not change much if we add an acid or a base).arrow_forward22-84 How many different dipeptides can be made (a) using only alanine, tryptophan, glutamic acid, and arginine and (b) using all 20 amino acids?arrow_forwardWhich of the following amino acids is the least polar? a) Fb) Dc) Hd) Tarrow_forward
- If your molecule C4H11N5 was the side chain of an amino acid, in a protein, what tertitary structure stabilizers could be present? Make a key and use colors to highlight what part of your structure can be stabilized in each manner?arrow_forwardWhat amino acid is found at this position: 148 of 4ALD? Explain why this position in your protein is important and outline the effects the mutation will have on the 3D structure and the function of your protein ( 4ALD).arrow_forwardAn amino acid contains by mass: 34.28% C, 6.71% H, 13.32% N, and 45.67% O. What is its empirical formula? C3H9N2O2 C2H5N2O3 C3H7NO3arrow_forward
- Which amino acids have R groups with an additional chiral carbon? - Cysteine - Isoleucine - Threonine -Histidine - Tryptophan - Tyrosine - Methioninearrow_forwardFor the tripeptide ala-lys-phe a) Draw the structure of the peptide you would expect at pH 1. Show all lone pairs of electrons and show formal charges. Determine the charge on the tripeptide. b) Draw the structure of the peptide you would expect at pH 7. Show all lone pairs of electrons and show formal charges. Determine the charge on the tripeptide..arrow_forwardThe amino acid alanine is a solid at room temperature and has a melting point of 315 °C, while pyruvic acid (CH 3COCO 2H) has a similar molecular weight but is a liquid at room temperature with a boiling point of 165 °C. Account for the difference.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning


