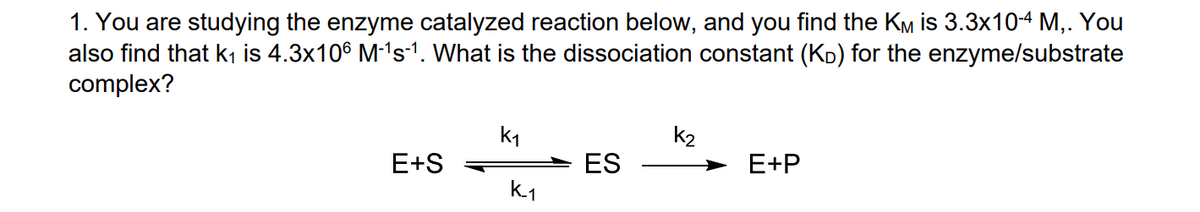
1. You are studying the enzyme catalyzed reaction below, and you find the KM is 3.3x104 M,. You also find that k1 is 4.3x106 M-'s-1. What is the dissociation constant (KD) for the enzyme/substrate complex? k1 k2 E+S ES E+P k1
Q: Which of the following statements are correct? explain your answers.a. The active site of an enzyme…
A: Explanation given for the statements.
Q: For the following reaction: k1 E+S ES P+E (a) What is the rate equation for the formation of ES? (b)…
A: Enzymes are biological catalysts that help in catalyzing or speeding up biological reactions by…
Q: The kinetics of an enzyme have been investigated experimentally. Some of the results are given in…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: You have discovered a new enzyme that has a nearly identical active site to chymotrypsin. This new…
A: As given peptide is Asn-Phe-lys, and discovered enzyme cleaves at C terminal of non ionizable polar…
Q: , non-ionizable R groups.
A: Introduction Beginning with the first tetrahedral middle of the road, draw the instrument of…
Q: Can you describe how electrostatic and steric considerations may lead to preferential stabilization…
A: Enzymes are proteinaceous molecules that catalyze a wide range of reactions that take place in…
Q: The AG of a reaction is -40 kcal/mol. The AG of the enzyme- catalyzed reaction is: Oa We cannot…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: An enzyme is present at a concentration of 1 nM and has a Vmax of 2 µM s-'. The Km for its primary…
A: K cat is nothing but turnover number of enzyme which is equal to number of substrate molecules…
Q: 4. An enzyme catalyzed reaction was initiated in 1.0 mL of 0.06 M HEPES buffer, pH 7.55. During the…
A: The Henderson Hasselbalch equation describes the mathematical relationship between the pH and pKa of…
Q: What is the difference between the left and right? What type of inhibition is being shown? What are…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: 1. What is the keat of this enzyme? 2. At which concentration of the substrate this enzyme reaches…
A: Given, Km = 200 μM [ETotal] = 10 nm = 0.01 Vmax = 2.5 μM/s
Q: 7. You obtain the following set of data for two enzymes that follow Michaelis-Menten kinetics:…
A: Michaelis Menton kinetics describes the relation between rate of reaction and substrate…
Q: Substrate Concentration [SI The graph above shows the initial rate of an enzyme-catalyzed reaction…
A: In the given question we are supposed to answer the connection between the enzyme primary structure…
Q: (a) At what substrate concentration would an enzyme with a kcat of 30.0s−1 and a Km of 0.0050 M…
A: The enzyme are the biocatalysts that increase the rate of reaction by decreasing the activation…
Q: Suppose you have 100 molecules of an enzyme (an aspartyl protease) that requires aspartate in the…
A: Pka of aminoacid is important to know the pH at which the Amino acid is protonated or deprotonated.…
Q: The key catalytic amino acids of the lysozyme active site in the free enzyme are Glu 35 with a…
A: The catalytic mechanism of lysozyme involves both general acid and general base catalysis.
Q: 2. Peter does not tolerate lactose in milk. He bought a solution of ß-galactosidase where [Eo] 10.0…
A: First lets find the Vmax value of the enzyme β-galactosidase. Vmax = Kcat×Etotal =525 s-1…
Q: 19. The picture below indicates a Lineweaver-Burk plot used to illustrate the values of KM and Vmax…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1. You are working on an experiment with a newly discovered enzyme. This enzyme works most…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: In enzyme and chemical kinetics A -->P, what is the instantaneous appearance or disappearance of A…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation energy…
Q: (Vmax.f [S] - (Vmax,b [P] KM,b [P] vo [S] 1+ KM.f KM.b ||
A: In this enzyme kinetics, I am going assume a general mechanism and then derive the rate equation. As…
Q: 2C Two homologues of this enzyme from different species both catalyze the reaction SY with distinct…
A: Hi! Thank you for the question. We are authorized to answer one question at a time, since you have…
Q: A research group discovers a new version of happyase, which they call happyase*, that catalyzes the…
A: Enzymes are usually protein molecules which increases the rate of biochemical reactions by…
Q: The following reaction coordinate diagram charts the energy of a substrate molecule (S) as it passes…
A: The catalysis of processes by enzymes is critical in living systems. Each organism goes through…
Q: In enzyme and chemical kinetics A -->P, what is the instantaneous appearance or disappearance of A…
A: Enzymes are protein molecules that increase the rate of the reaction by decreasing the activation…
Q: 1. Define the following: Active site Substrate • Enzyme-substrate complex • Rate of reaction 2. What…
A: Active site:- In an enzyme, that area where the substrate molecules bind and undergo a chemical…
Q: Curve N represents the curve for an allosteric enzyme with no allosteric activators or inhibitors…
A: Allosteric enzymes have multiple active sites and also multiple subunits. Thus the graph obtained is…
Q: You have discovered a new enzyme that has a nearly identical active site to chymotrypsin. This new…
A: It is mentioned that the newly discovered enzyme with a reaction mechanism similar to that of the…
Q: When you plot [S] versus initial velocity for an allosteric enzyme, what type of plot arises: *…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation energy…
Q: An enzyme catalyzes a reaction in which substrateA is cleaved into two products, P and Q. In the…
A: A competitive inhibitor binds to active site of an enzyme and prevents the binding of actual…
Q: 1. The following data were recorded for the enzyme-catalyzed reaction S → P. [S] in M Imole/min…
A: The rate of a biochemical reaction is increased by the proteins known as enzymes. Enzymes increase…
Q: Which of the following statements about a plot of V0 vs. [S] for an enzyme that follows…
A: Michaelis-Menten kinetics equation Vo = Vmax[S]/(Km+[S]) Vo is reaction rate Vmax is maximum…
Q: An enzyme catalyzes a reaction with a Km of 8.50 mM and a Vmax of 3.45 mM · s-. Calculate the…
A: Given Values: Km = 8.5 mM Vmax = 3.45 mM s-1
Q: H2O2 + catalase = 2 H2O2 + O2, is an example of what enzyme specificity? * A. Stereochemical B.…
A: Catalase is an enzyme seen in all living organisms and they are responsible for the neutralization…
Q: You have obtained experimental kinetic data for two versions of the same enzyme, a wild‑type and a…
A: Michaelis-Mention constant (Km) or equilibrium constant for the dissociation of the E-S complex is…
Q: a. Is catalase activity endothermic or exothermic? b. What classification of enzyme is catalase?…
A: Catalase is an enzyme that catalyzes the breakdown of hydrogen peroxide into water and oxygen. It is…
Q: In a bisubstrate reaction, a small amount of the fi rst product P is isotopicallylabeled (P*) and…
A: The Ping-Pong mechanism is a non-sequential mechanism. A product is released after the first…
Q: Shown below is a hypothetical reaction pathway where both E and H can be produced from compound A.…
A: Enzymes are specialized proteins that catalyze all biochemical reactions. An enzyme can be regulated…
Q: The Vmax for a particular enzyme is 10 nmols/L/s. The Km for its substrate is 5 microM. If the…
A: Given that, the Vmax for a particular enzyme is 10 nmol/L/s. The Km for its substrate is 5 uM. We…
Q: A research group discovers a new version of happyase, that catalyzes the chemical reaction HAPPY…
A: An enzyme can be used repeatedly to convert substrates into products. Each has a particular capacity…
Q: An enzyme E binds a substrate S and a cofactor C. The equilibrium dissociation constant Kd,S of the…
A: Enzymes are usually made of protein molecules which increase the rate of biochemical reaction by…
Q: 1.The kinetics of an enzyme are measured as a function of substrate concentration in the presence or…
A: An enzyme inhibitor is a molecule that binds to an enzyme and decreases its activity or rate of…
Q: 5. For a Michaelis-Menten enzyme, k1 = 5.2 ⅹ 108 M-1 s-1, k-1 = 3.1 ⅹ 104 s-1, and k2 = 3.4 ⅹ 105…
A: Michaelis-Menten model of enzyme kinetics is a very useful model which describe the relationship…
Q: A research group discovers a new version of happyase, which they call happyase*, that catalyzes the…
A: Enzymes are specialized proteins that catalyze all biochemical reactions. Enzymes are divided into…
Q: From the Lineweaver-Burke plot of an enzyme-catalyzed reaction containing 4 μM total enzyme, you…
A: Hi. Thank you for the question. As per the honor code, We'll answer the first question since the…
Q: Under conditions where this enzyme is saturated, which parameter will change significantly as you…
A: The Michaelis-Menten (MM) model is the one of the simplest approaches to enzyme kinetics. MM…
Q: An enzyme contains an active site aspartic acid with a pKa = 5.0, whichacts as a general acid…
A: Enzymes contain ionizable groups at the active site for general acid catalysis. The activity of the…
Q: 1. A reaction in which a substrate is converted to a product is catalyzed by an enzyme. The reaction…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: 1. The concentration of substrate X is high. What happens to the rate of the enzyme-catalyzed…
A: Enzymes are the proteinaceous substances that are capable of catalyzing chemical reactions of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- At what substrate concentration would an enzyme with a kcat of 25.0 s-1 and a KM of 3.5 mM operate at 25% of its maximal rate? How many reactions would the enzyme catalyze in 45 seconds when it is fully saturated with substate, assuming the enzyme has one active site?1. Can you describe how electrostatic and steric considerations may lead to preferential stabilization of the transition state at an enzyme active site? 2. What factors are involved in “transition-state complementarity”?1. The concentration of substrate X is high. What happens to the rate of the enzyme-catalyzed reaction if the concentration of substrate X is reduced? Explain. 2. An enzyme has an optimum pH of 7.2. What is most likely to happen to the activity of the enzyme if the pH drops to 6.2? Explain
- Enzyme A catalyzes the reaction S → P and has a KM of 50 μM and a Vmax of 100 nM s–1. EnzymeB catalyzes the reaction S → Q and has a KM of 5 mM and a Vmax of 120 nM s–1. When 100 μM ofS is added to a mixture containing equal amounts of enzymes A and B, which reaction product (Por Q) will be more abundant after 1 minute of reaction?A research group discovers a new version of happyase, which they call happyase*, that catalyzes the chemical reaction The researchers begin to characterize the enzyme. (a) In the first experiment, with [Et] at 4 nM, they find that the Vmax is 1.6 uM/s. Based on this experiment, what is the kcat for happyase*? (Include appropriate units.) (b) In another experiment, with [Et] at 1 nM and [HAPPY] at 30 uM, the researchers find that V0 = 300 nM/s. What is the measured Km of happyase* for its substrate HAPPY? (Include appropriate units.) (c) Further research shows that the purified happyase* used in the first two experiments was actually contaminated with a reversible inhibitor called ANGER. When ANGER is carefully removed from the happyase* preparation and the two experiments repeated, the measured Vmax in (a) is increased to 4.8 uM/s, and the measured Km in (b) is now 15 uM. Based on this information, can you figure out what type of inhibitor is ANGER? (Use table 6.9 at the end of the…An enzyme catalysed reaction has a Km of 8 mM and a Vmax of 13 nM.s-1. Use the Michaelis-Menten equation to calculate the reaction velocity when the substrate concentration is 18 mM.
- a. What is the Vmax of this enzyme WITHOUT inhibitor? Please show your work. b. What is the Km of this enzyme WITHOUT inhibitor? Please show your work. c. The specificity constant of enzyme X is 8 x 10^7 /(M * seconds) What is the kcat of enzyme X WITHOUT inhibitor? Please show your work d. What was the concentration of enzyme used for measuring the kinetics of enzyme X WITHOUT inhibitor? Please show your work3.18: An enzyme E binds a substrate S and a cofactor C. The equilibrium dissociation constantKd,S of the enzyme-substrate complex ES is 1 μM, for EC it is 10 μM. When the cofactor Cis present, K’d,S is decreased to 0.1 μM. What is the value for the dissociation constant K’d,C of the enzyme-cofactor complex in the presence of substrate S? Calculate the interactionenergy ΔΔGint for cofactor and substrate binding.1.1)the following data duscribe an enzyme-catalyzed reaction(hydrolysis of cabobenzoxyglycyl-L-tryptophan) Plot these results using a lineweaver-Burk method, and determine values for Km and Vmax. substrate concenrate(mM) Velocity(mM.sec-1) 2,5 0.024 5 0.036 10 0.053 15 0.060 20 0.061 25 0.062 1.2) If the Km of an enzyme for it's substrate remains constant as the concentration of the inhibitor icreaces, what can be said about the mode of inhibition and why? 1.3) calculate the turnover number for an enzyme, assuming Vmax is 0.5M.sec-1 and the concentration of the enzyme used is 0.002M . why is it usefull to know this? 1.4) discuss the mechanism of the bohr effect that occurs during the interactions of Hb with oxygen under physiological conditions in the lungs and tissues. make use of relavant graphs and diagrams to explain your answer.
- In a bisubstrate reaction, a small amount of the fi rst product P is isotopicallylabeled (P*) and added to the enzyme and the fi rst substrate A. No B or Q is present. Will A (= P—X) become isotopically labeled (A*) if the reaction follows a Ping Pong mechanism?An enzyme catalyzes a reaction in which substrate A is cleaved into two products, P and Q. In the catalytic mechanism, the enzyme converts A to an covalently-bound reaction intermediate X and product P, P then desorbs from the enzyme, and in a second chemical step, the enzyme converts the intermediate X in the EX complex to the final product Q (in EQ), which then desorbs from the enzyme E. You discover two inhibitors of this enzyme, I and J. I is a competitive inhibitor of the substrate A, and has nearly double the molecular weight of J. On the other hand, J is a mixed inhibitor of enzyme E, and its inhibitory effect on Km / Vmax (the slope effect from the double reciprocal plot) is greater than that of 1 / Vmax (the intercept effect in a double reciprocal plot). That is Kis < Kii . At low pH, the conversion of EX to EQ is greatly slowed, kcat is decreased, and the intercept effect of inhibitor J is elevated, that is, the value of Kii is diminished. When a high, fixed concentration…A particular enzyme-catalyzed reaction has an apparent Vmax = 9.00 nmol s-1 and α' = 3.00 when 2.00 µmol L-1 inhibitor X is present and uncompetitively inhibiting the reaction. Calculate Vmax for the uninhibited reaction in nmol s-1.

