1.008 4. He 4.003 10 Be 6.941 9.012 LI B C N 10.81 12.01 14.01 16.00 19.00 20.18 15 F Ne 11 12 13 14 16 17 18 Mg 22.99 2431 Na Al Si P Ar 2698 28.09 30.97 32.07 35 45 39.95 31 19 20 21 22 23 24 25 Mn 26 27 28 29 30 32 33 34 35 36 Ca Se Ti Cr Fe Zn Co NI 39.10 40 08 44 96 47 88 50 94 52 00 54.94 55 85 58 93 58 69 63 55 65 38 69.72 72.59 74.92 78 96 79,90 83.80 Cu Ga Ge As Se Br Kr 37 38 39 40 41 42 43 44 45 46 47 48 54 49 30 52 53 Rb Sr Zr Nb Mo Te Ru Rh Pd 85.47 87 62 88 9191 22 9291 95 94 (98) 1011 102 9 106 4 10 9 11241148118 71218 1276 126 9 131 3 Ag Cd In Sn Sb Te Xe 55 56 57 72 73 74 75 76 77 78 79 S1 $2 SO Hg 200 6 204 4 207 2 209.0 (209) (210)|(222) $3 84 $6 Cs Ba La Ta 1329 1373138 9|178 S|180 9 183.9|186 | 190 2|192 WE Re Os Ir Pt Au 19 TI Pb Bi Po At Ra 195 89 Ac (2233 226 (227) 87 Fr 8S Ra Question 7/10 ميز هذا السؤال A 0.500 g of KCl was dissolve in 100 mL volumetric flask, and 10 mL from this solution was pipette and titrations against 7.50 mL of silver nitrate solution, the molariry of silver nitrate solution is: ( the molar mass of KCI = 74.5 g mol) 1. O 0.089 M. 2. 00.054 M. 3.00.064 M. 4.00.071 M. 0.023 M.
1.008 4. He 4.003 10 Be 6.941 9.012 LI B C N 10.81 12.01 14.01 16.00 19.00 20.18 15 F Ne 11 12 13 14 16 17 18 Mg 22.99 2431 Na Al Si P Ar 2698 28.09 30.97 32.07 35 45 39.95 31 19 20 21 22 23 24 25 Mn 26 27 28 29 30 32 33 34 35 36 Ca Se Ti Cr Fe Zn Co NI 39.10 40 08 44 96 47 88 50 94 52 00 54.94 55 85 58 93 58 69 63 55 65 38 69.72 72.59 74.92 78 96 79,90 83.80 Cu Ga Ge As Se Br Kr 37 38 39 40 41 42 43 44 45 46 47 48 54 49 30 52 53 Rb Sr Zr Nb Mo Te Ru Rh Pd 85.47 87 62 88 9191 22 9291 95 94 (98) 1011 102 9 106 4 10 9 11241148118 71218 1276 126 9 131 3 Ag Cd In Sn Sb Te Xe 55 56 57 72 73 74 75 76 77 78 79 S1 $2 SO Hg 200 6 204 4 207 2 209.0 (209) (210)|(222) $3 84 $6 Cs Ba La Ta 1329 1373138 9|178 S|180 9 183.9|186 | 190 2|192 WE Re Os Ir Pt Au 19 TI Pb Bi Po At Ra 195 89 Ac (2233 226 (227) 87 Fr 8S Ra Question 7/10 ميز هذا السؤال A 0.500 g of KCl was dissolve in 100 mL volumetric flask, and 10 mL from this solution was pipette and titrations against 7.50 mL of silver nitrate solution, the molariry of silver nitrate solution is: ( the molar mass of KCI = 74.5 g mol) 1. O 0.089 M. 2. 00.054 M. 3.00.064 M. 4.00.071 M. 0.023 M.
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter1: Basic Concepts Of Chemistry
Section: Chapter Questions
Problem 43GQ: Hexane (C6H14, density = 0.766 g/cm3), perfluoro-hexane (C6F14, density = 1.669 g/cm3), and water...
Related questions
Question
Transcribed Image Text:1.008
4.
He
4.003
3.
10
Be
6.941 9.012
LI
B
N
10.81 12.01 14.01 16.00 19.00 20.18
15
Ne
11
12
13
14
SI
16
17
18
Na
Mg
Al
P
Ar
22.99 24.31
2698 28.09 30.97 32.07 35 4539 95
19
20
21
22
23
24
25
Mn
26
27
31
30
Zn
39.10 40 08 44.96 47 88 5094 52 00 54.9455.85 S8 93 58 69 63 55 65 38 69.
32
Ga
28
29
33
34 35
36
Ca
Se TI
Cr
Fe
Co
NI
Cu
Ge
72.59 74.92 78 96 79.90 83.80
49
As
Se
Br
Kr
37 38
39
40
41
42
43
44
45
46
47
48
50
51
52
53
54
Rb Sr Y
Zr
Nb Mo
Te
Ru
Rh
Pd
85.47 87 62 88 9191 22 9291 95 94 (98) 1011 102 9 106 4 10 9 11241148 118.7121 8 1276 126.9 131 3
Ag
Cd
In
Sn
Sb
Te
Xe
55
56
57 72
73
74
75
76
77
78
79
$2
$6
Ra
SO
81
$3
84
Cs
Ba
La
Re
Ta
132-9 1373138 9|178 S|180 9 183.9|186 190 2|192
WE
Os
Ir
Pt
Au
19
Hg
TI
Pb
Bi
Po
At
195
200 6 204 4 207.2 209.0|(209) (210) 1222)
89
Ae
(2233 226 (227)
87
Fr
Ra
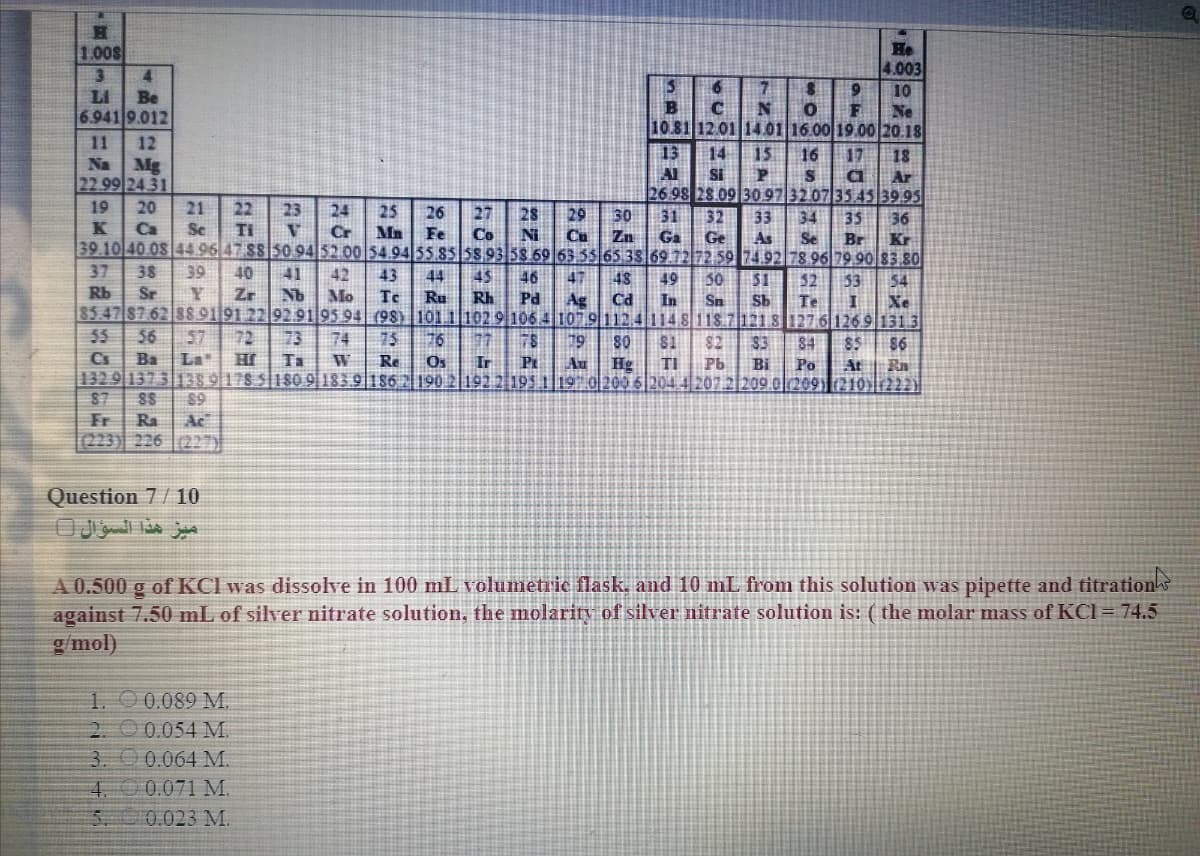
Question 7/10
ميز هذا السؤال ل
A 0.500 g of KCI was dissolve in 100 mL volumetric flask, and 10 mL from this solution was pipette and titrations
against 7.50 mL of silver nitrate solution, the molariry of silver nitrate solution is: ( the molar mass of KCl = 74.5
g/mol)
1. O 0.089 M.
2. O 0.054 M.
3. 00.064 M.
4. 0 0.071 M.
0.023 M.
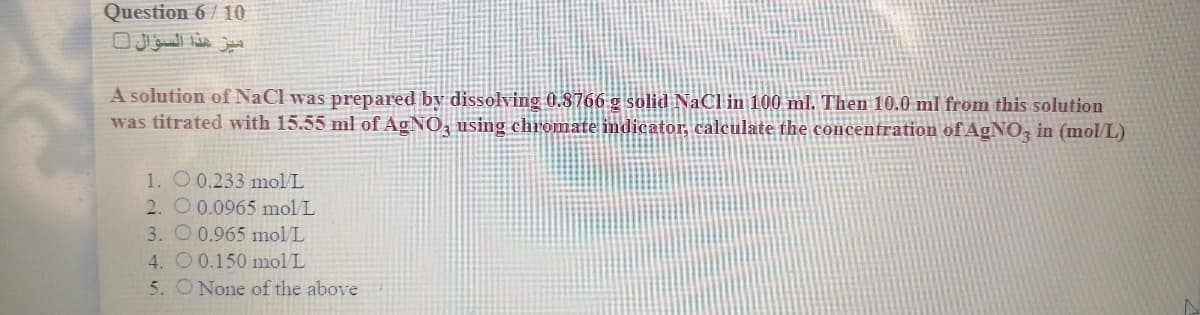
Transcribed Image Text:Question 6 / 10
A solution of NaCl was prepared by dissolving 0.8766 g solid NaCl in 100 ml. Then 10.0 ml from this solution
was titrated with 15.55 ml of AGNO, using chromate indicator, calculate the concentration of AGNO, in (mol/L)
1. O 0.233 molL
2. O 0.0965 mol L
3. O 0.965 mol L
4. O0.150 mol/L
5. O None of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co
