
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Transcribed Image Text:9 Schoology
9 Grades | Schoology
9 Schoology
9 Grades | Schoology
A bcps.schoology.com/common-assessment-delivery/start/4509684158?action=Donresume&submissionld=3783873-
E Apps
Downloading SIMS.
M Gmail
O YouTube A Maps D Watch Disney Chan..
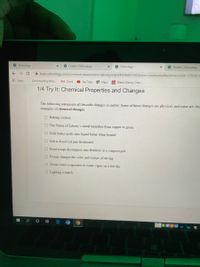
1/4 Try It: Chemical Properties and Changes
The following statements all describe changes in matter. Some of these changes are physical, and some are che
examples of chemical changes.
O Baking cookies
O The Statue of Liberty's metal tarnishes from copper to green
O Cold butter melts into liquid butter when heated
O Salt is dissolved into freshwater
O Food scraps decompose into fertilizer in a compost pile
O Frying changes the color and texture of an egg
O Ocean water evaporates to water vapor on a hot day
O Lighting a match
立
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of th following are chemical changes? and which are physical changes? making syrup by heating maple sap to remove water through evaporaiton?arrow_forwardDetermine whether each change is physical or chemical.arrow_forwardA student poured two liquids (ethylene glycol and mercury) into a tall graduated cylinder to perform an experiment. Which of the following observations is false? Iron Density of various substances Substance Ethylene glycol Chloroform Mercury Gold Density (g/cm³) 7.8 1.09 1.49 13.5 19.32 O Chloroform floats on top of ethylene glycol. O Iron floats on top of mercury. O Iron sinks in chloroform. O Gold sinks in mercury. O Ethylene glycol is on the topmost layer.arrow_forward
- Identify the physical property listed O Dynamite explodes O Sulfuric acid is corrosive O None of the listed are a physical property O Gasoline is flammable O Boiling an eggarrow_forwardAt room temperature, nitrogen does not have a definite shape or volume. Which state is it In? water gas O solid liquid There are three main states of matter: solid liguid and gas All have different physical nroperties Which state NC TEST DELL & 4. 7 S C %# 3arrow_forwardAt room temperature and pressure, classify each of the following macroscopic samples as solid, liquid or gas: salt water nitrogen O Use the References to access important values if needed for this question. 0 In the following descriptions the particles are atoms, molecules, or ions. To which sample above does each description best apply? The particles are in close contact and are held in place. The spaces between the particles is the largest. Carrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY