14. Here is shown a plot of the logarithm (to the base 10) of the diffusion coefficient versus reciprocal of the absolute temperature, for the diffusion of 10-15 iron in chromium. Determine values for the activation energy and preexponential. 10-16 0.55 0.60 0.65 0.70 Reciprocal temperature (1000/K) Diffusion coefficient (m²/s)
14. Here is shown a plot of the logarithm (to the base 10) of the diffusion coefficient versus reciprocal of the absolute temperature, for the diffusion of 10-15 iron in chromium. Determine values for the activation energy and preexponential. 10-16 0.55 0.60 0.65 0.70 Reciprocal temperature (1000/K) Diffusion coefficient (m²/s)
Chapter34: Miscellaneous Separation Methods
Section: Chapter Questions
Problem 34.19QAP
Related questions
Question
14
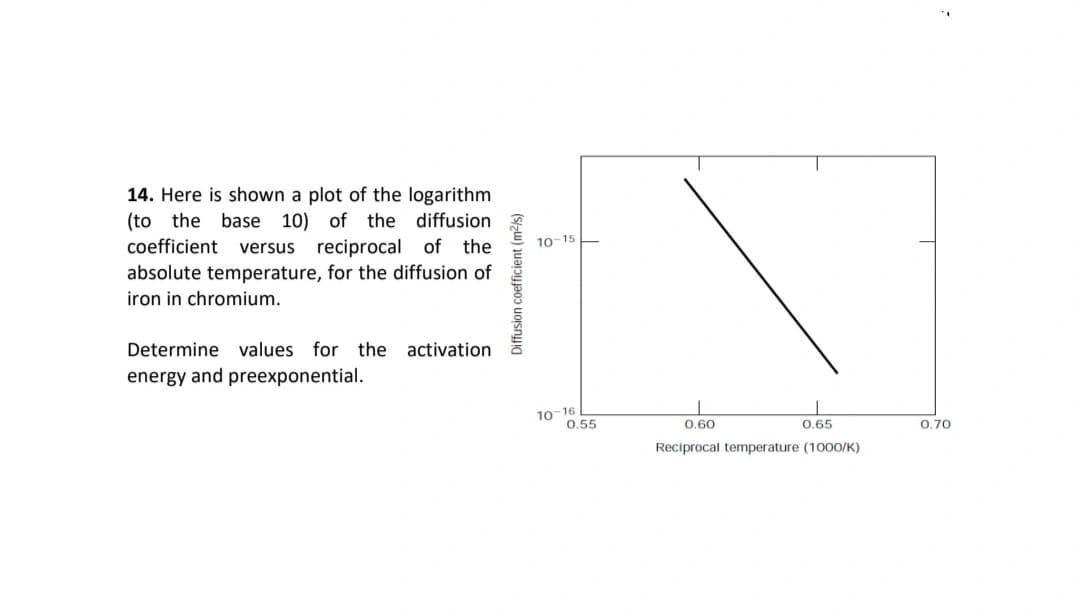
Transcribed Image Text:14. Here is shown a plot of the logarithm
(to the base 10) of the diffusion
coefficient
10-15
versus reciprocal of the
absolute temperature, for the diffusion of
iron in chromium.
Determine values for the activation
energy and preexponential.
10-16
0.55
0.60
0.65
0.70
Reciprocal temperature (1000/K)
Diffusion coefficient (m²/s)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 5 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

