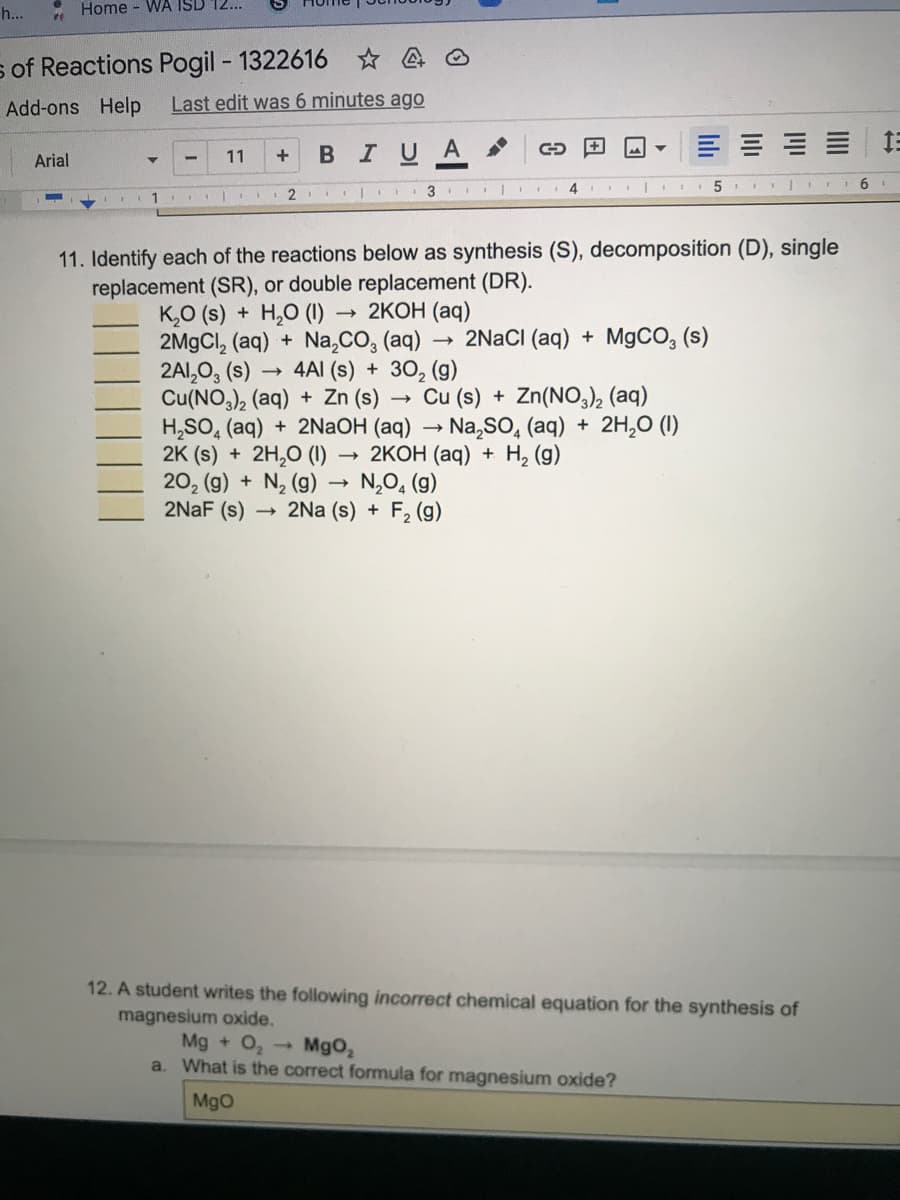
2 3 11. Identify each of the reactions below as synthesis (S), decomposition (D), single replacement (SR), or double replacement (DR). K,O (s) + H,O (1) 2M9CI, (aq) + Na,CO, (aq) 2AI,0, (s) Cu(NO,), (aq) + Zn (s) H,SO, (aq) + 2NAOH (aq) Na,So, (aq) + 2H,0 (I0) 2K (s) + 2H,0 (1) → 2KOH (aq) + H, (g) 20, (g) + N, (9) 2NAF (s) 2KOH (aq) 2NaCI (aq) + M9CO, (s) - 4AI (s) + 3O, (g) Cu (s) + Zn(NO,), (aq) → N,O, (g) - 2Na (s) + F, (g)
2 3 11. Identify each of the reactions below as synthesis (S), decomposition (D), single replacement (SR), or double replacement (DR). K,O (s) + H,O (1) 2M9CI, (aq) + Na,CO, (aq) 2AI,0, (s) Cu(NO,), (aq) + Zn (s) H,SO, (aq) + 2NAOH (aq) Na,So, (aq) + 2H,0 (I0) 2K (s) + 2H,0 (1) → 2KOH (aq) + H, (g) 20, (g) + N, (9) 2NAF (s) 2KOH (aq) 2NaCI (aq) + M9CO, (s) - 4AI (s) + 3O, (g) Cu (s) + Zn(NO,), (aq) → N,O, (g) - 2Na (s) + F, (g)
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
I need help with this question
(Not honor class)
(Not grading)
Transcribed Image Text:h...
Home - WA ISD 12...
s of Reactions Pogil - 1322616 ☆ 4
Add-ons Help
Last edit was 6 minutes ago
11
IUA
Arial
4.
6.
1
11. Identify each of the reactions below as synthesis (S), decomposition (D), single
replacement (SR), or double replacement (DR).
K,O (s) + H,0 (1)
2M9CI, (aq) + Na,CO, (aq)
2AI,0, (s)
Cu(NO,), (aq) + Zn (s)
H,SO, (aq) + 2NaOH (aq) → Na,SO, (aq) + 2H,0 (1)
2K (s) + 2H,0 (1)
20, (g) + N, (g) → N,0, (g)
2NAF (s)
2КОН (аq)
2NACI (aq) + M9CO, (s)
4AI (s) + 30, (g)
Cu (s) + Zn(NO), (aq)
2КОН (ад) + Н, (9)
2Na (s) + F2 (g)
12. A student writes the following incorrect chemical equation for the synthesis of
magnesium oxide.
Mg + O, -
a. What is the correct formula for magnesium oxide?
Mgo,
MgO
III
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

