-2 Sio Si Fa 4 mg 7g 9. 12:17 AM Thu Dec 5 Done Done AA e www-awu.ale s.com O STOICHIOMETRY Limiting reactants Jaquesha Gaseous ethane (CH,CH,) will react with gaseous oxygen (02) to produce gaseous carbon dioxide (CO2) and gaseous water (H,0). Suppose 21. g of ethane is mixed with 157.g of oxygen. Calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. Be sure your answer has the correct number of significant digits. x10
-2 Sio Si Fa 4 mg 7g 9. 12:17 AM Thu Dec 5 Done Done AA e www-awu.ale s.com O STOICHIOMETRY Limiting reactants Jaquesha Gaseous ethane (CH,CH,) will react with gaseous oxygen (02) to produce gaseous carbon dioxide (CO2) and gaseous water (H,0). Suppose 21. g of ethane is mixed with 157.g of oxygen. Calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. Be sure your answer has the correct number of significant digits. x10
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 32QAP: When yeast is added to a solution of glucose or fructose, the sugars are said to undergo...
Related questions
Question
How do I solve?
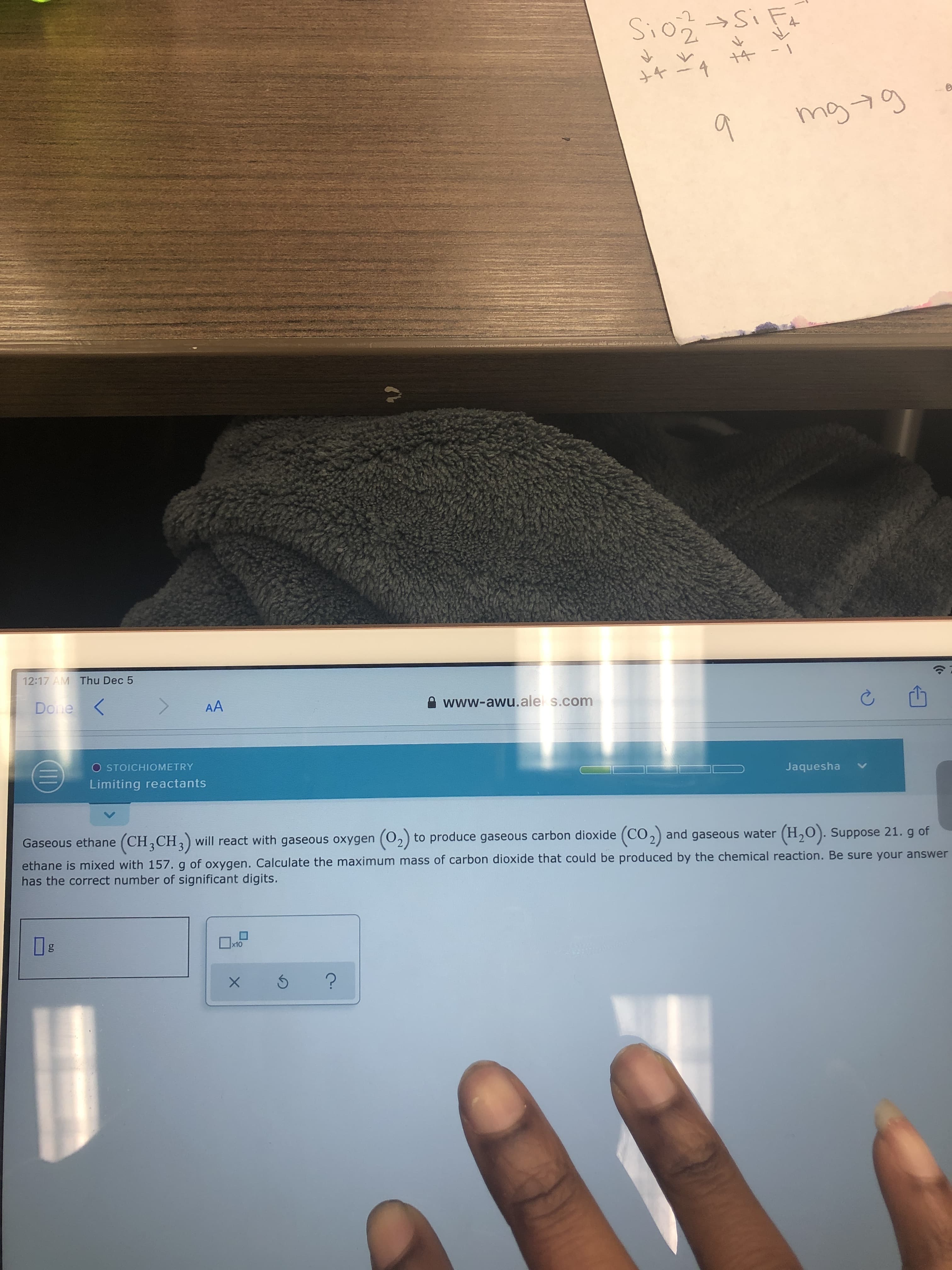
Transcribed Image Text:-2
Sio Si Fa
4
mg 7g
9.
12:17 AM Thu Dec 5
Done
Done
AA
e www-awu.ale s.com
O STOICHIOMETRY
Limiting reactants
Jaquesha
Gaseous ethane (CH,CH,) will react with gaseous oxygen (02) to produce gaseous carbon dioxide (CO2) and gaseous water (H,0). Suppose 21. g of
ethane is mixed with 157.g of oxygen. Calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. Be sure your answer
has the correct number of significant digits.
x10
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning