2) Suppose that we have 100 cm' of water (p = 1g.cm"), which is D;0 in a proportion of 0.015% (D stands for deuterium, or H). a) Compute the energy (in J) that would obtain from this sample if all the deuterium were to be consumed in the reaction H + H- 'H +p, which is known to release an energy output of 4.0 MeV. b) As an alternative, compute the energy (in J) that would be released if two-thirds of the deuterium were fused (according to the previous reaction) to form 'H, which then combines with the remaining one third according to the reaction H+ 'H → *He +n, known to release an energy output of 17.6 MeV. c) Which of the processes a) or b) produces more energy? Given: N= 6.022x10" mot P. ->
2) Suppose that we have 100 cm' of water (p = 1g.cm"), which is D;0 in a proportion of 0.015% (D stands for deuterium, or H). a) Compute the energy (in J) that would obtain from this sample if all the deuterium were to be consumed in the reaction H + H- 'H +p, which is known to release an energy output of 4.0 MeV. b) As an alternative, compute the energy (in J) that would be released if two-thirds of the deuterium were fused (according to the previous reaction) to form 'H, which then combines with the remaining one third according to the reaction H+ 'H → *He +n, known to release an energy output of 17.6 MeV. c) Which of the processes a) or b) produces more energy? Given: N= 6.022x10" mot P. ->
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter11: Rate Of Reaction
Section: Chapter Questions
Problem 99QAP: The gas-phase reaction between hydrogen and iodine H2(g)+I22HI(g)proceeds with a rate constant for...
Related questions
Question
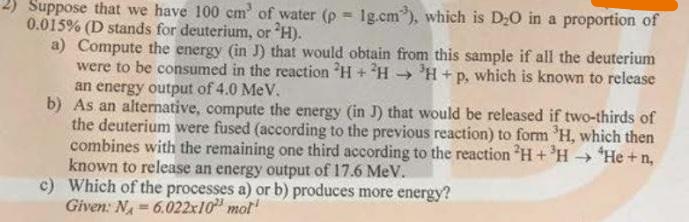
Transcribed Image Text:2) Suppose that we have 100 cm' of water (p = 1g.cm"), which is D;0 in a proportion of
%3!
0.015% (D stands for deuterium, or H).
a) Compute the energy (in J) that would obtain from this sample if all the deuterium
were to be consumed in the reaction H + H - 'H+p, which is known to release
an energy output of 4.0 MeV.
b) As an alternative, compute the energy (in J) that would be released if two-thirds of
the deuterium were fused (according to the previous reaction) to form 'H, which then
combines with the remaining one third according to the reaction H+ 'H → *He +n,
known to release an energy output of 17.6 MeV.
c) Which of the processes a) or b) produces more energy?
Given: N = 6.022x10" mot
P.
->
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,