21. What must also be present in the Mix of starch and salt. 22. What can you infer about whethe chip? Explain your reasoning. 23. Use your answers to Questions 1
21. What must also be present in the Mix of starch and salt. 22. What can you infer about whethe chip? Explain your reasoning. 23. Use your answers to Questions 1
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 1P
Related questions
Question
Answer question 21 and 22 short answers
Transcribed Image Text:monica. mnt..
TOU TUD
d-ons Help Accessibility
Last edit was 4 hours ago
Open Sans
BIUA
1 1
12
3. I.. 4
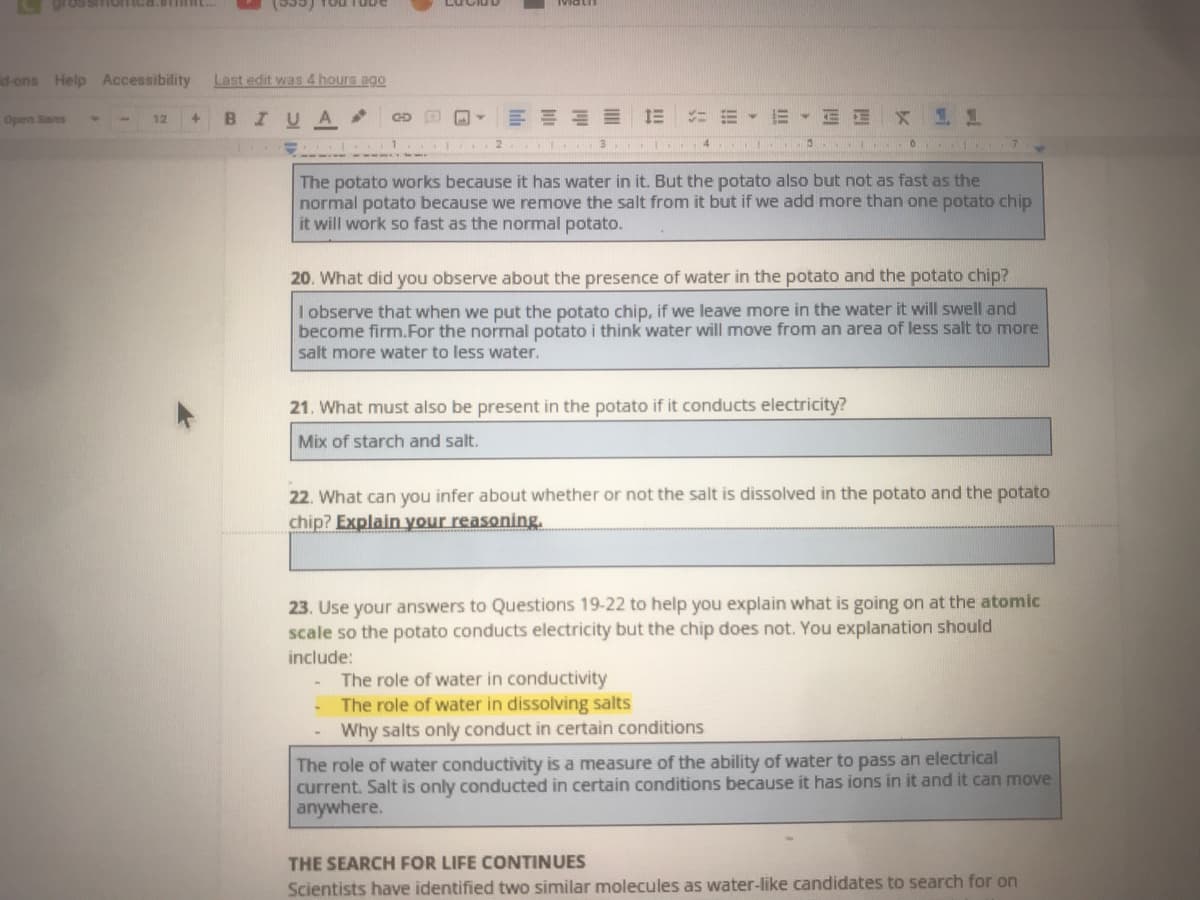
The potato works because it has water in it. But the potato also but not as fast as the
normal potato because we remove the salt from it but if we add more than one potato chip
it will work so fast as the normal potato.
20. What did you observe about the presence of water in the potato and the potato chip?
I observe that when we put the potato chip, if we leave more in the water it will swell and
become firm.For the normal potato i think water will move from an area of less salt to more
salt more water to less water.
21. What must also be present in the potato if it conducts electricity?
Mix of starch and salt.
22. What can you infer about whether or not the salt is dissolved in the potato and the potato
chip? Explain your reasoning.
23. Use your answers to Questions 19-22 to help you explain what is going on at the atomic
scale so the potato conducts electricity but the chip does not. You explanation should
include:
The role of water in conductivity
The role of water in dissolving salts
Why salts only conduct in certain conditions
The role of water conductivity is a measure of the ability of water to pass an electrical
current. Salt is only conducted in certain conditions because it has ions in it and it can move
anywhere.
THE SEARCH FOR LIFE CONTINUES
Scientists have identified two similar molecules as water-like candidates to search for on
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning