Chapter96: Carbon-13 Nuclear Magnetic Resonance Spectroscopy
Section: Chapter Questions
Problem 2P
Related questions
Question
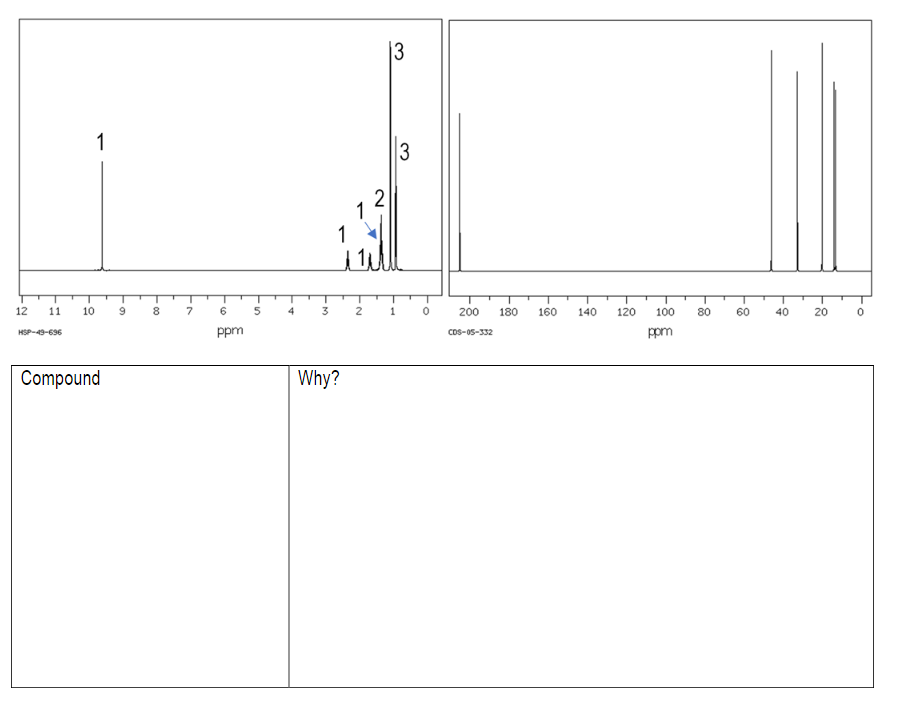
Transcribed Image Text:3
1
2
1
12
11
10
8
7
6
2
200
180
160
140
120
100
80
60
40
20
HSP-49-696
ppm
Cos-05-332
ppm
Compound
Why?
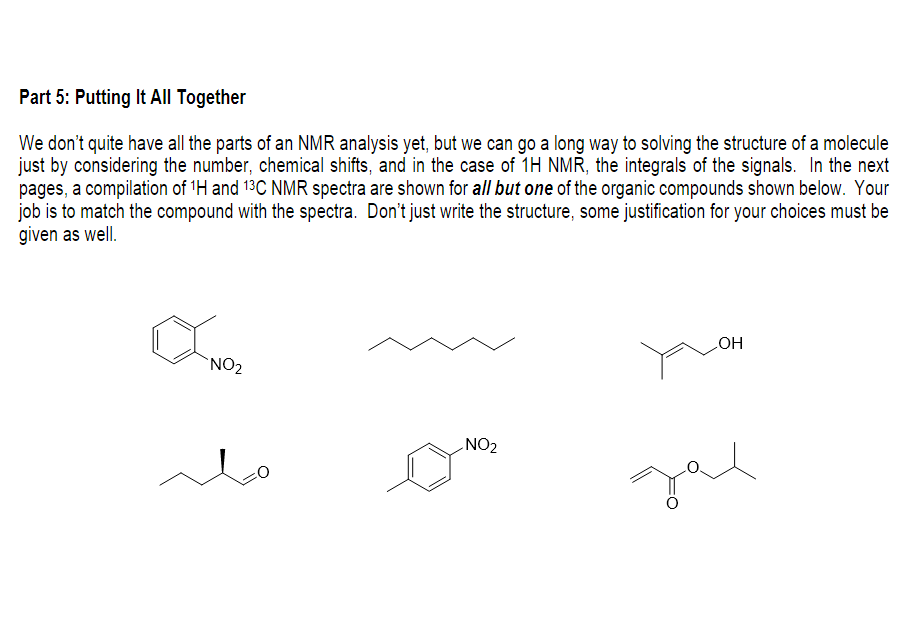
Transcribed Image Text:Part 5: Putting It All Together
We don't quite have all the parts of an NMR analysis yet, but we can go a long way to solving the structure of a molecule
just by considering the number, chemical shifts, and in the case of 1H NMR, the integrals of the signals. In the next
pages, a compilation of 'H and 13C NMR spectra are shown for all but one of the organic compounds shown below. Your
job is to match the compound with the spectra. Don't just write the structure, some justification for your choices must be
given as well.
`NO2
HO
NO2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning