3. Which of these elements would you expect to form the following with copper: Electro- Atomic Radius Crystal Structure nega- tivity (a) A substitutional solid solution having complete solubility? Element Valence (nm) 0.1278 Cu FCC 1.9 +2 C 0.071 0.046 0.060 0.1445 H (b) A substitutional solid solution of incomplete Ag AI FCC FCC 1.9 +1 +3 solubility? 0.1431 1.5 Co 0.1253 НСР 18 +2 +3 0.1249 ВСС 1.6 (c) An interstitial solid solution? Fe NI Pd 0.1241 0.1246 ВСС FCC FCC 1.8 +2 1.8 +2 0.1376 2.2 +2 Pt Zn FCC НСР 0.1387 2.2 +2 0.1332 1.6 +2
3. Which of these elements would you expect to form the following with copper: Electro- Atomic Radius Crystal Structure nega- tivity (a) A substitutional solid solution having complete solubility? Element Valence (nm) 0.1278 Cu FCC 1.9 +2 C 0.071 0.046 0.060 0.1445 H (b) A substitutional solid solution of incomplete Ag AI FCC FCC 1.9 +1 +3 solubility? 0.1431 1.5 Co 0.1253 НСР 18 +2 +3 0.1249 ВСС 1.6 (c) An interstitial solid solution? Fe NI Pd 0.1241 0.1246 ВСС FCC FCC 1.8 +2 1.8 +2 0.1376 2.2 +2 Pt Zn FCC НСР 0.1387 2.2 +2 0.1332 1.6 +2
Chapter4: Calculations Used In Analytical Chemistry
Section: Chapter Questions
Problem 4.32QAP
Related questions
Question
100%
33
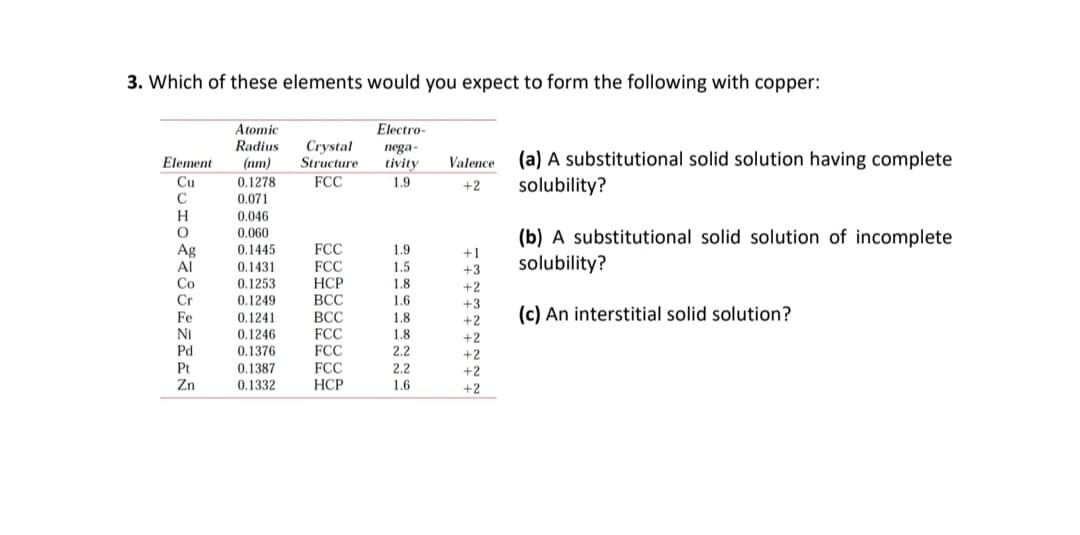
Transcribed Image Text:3. Which of these elements would you expect to form the following with copper:
Atomic
Radius
Electro-
Crystal
Structure
nega-
(a) A substitutional solid solution having complete
solubility?
Element
(nm)
tivity
Valence
Cu
FCC
0.1278
0.071
1.9
+2
H
0.046
0.060
0.1445
(b) A substitutional solid solution of incomplete
FCC
Ag
Al
Co
Cr
1.9
+1
solubility?
0.1431
0.1253
0.1249
FCC
1.5
+3
НСР
ВСС
1.8
+2
+3
1.6
Fe
(c) An interstitial solid solution?
0.1241
0.1246
ВСС
FCC
1.8
+2
NI
1.8
+2
Pd
0.1376
FCC
2.2
+2
Pt
Zn
FCC
НСР
0.1387
2.2
+2
0.1332
1.6
+2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning