3A. For the following tripeptide, provide its full name, abbreviated name, describe what type of interactions would be present in the tertiary structure of a protein that contains this sequence. In other words, evaluate what types of interactions can occur between the amino acids provided in this peptide and the functional groups in different amino acid side chains. How would this peptide interact with water? Explain why Full Name: Abbreviated Name: Interactions: Interactions with water: H. H3N-CHC-N-CHC-N-CHC-O CH2 CH2 CH2 CH2 O NH3 CH2 CH2 OH NH
3A. For the following tripeptide, provide its full name, abbreviated name, describe what type of interactions would be present in the tertiary structure of a protein that contains this sequence. In other words, evaluate what types of interactions can occur between the amino acids provided in this peptide and the functional groups in different amino acid side chains. How would this peptide interact with water? Explain why Full Name: Abbreviated Name: Interactions: Interactions with water: H. H3N-CHC-N-CHC-N-CHC-O CH2 CH2 CH2 CH2 O NH3 CH2 CH2 OH NH
Biology 2e
2nd Edition
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:Matthew Douglas, Jung Choi, Mary Ann Clark
Chapter3: Biological Macromolecules
Section: Chapter Questions
Problem 23CTQ: Amino acids have the generic structure seen below, where R represents different carbon-based side...
Related questions
Question
Help please. Thank you!
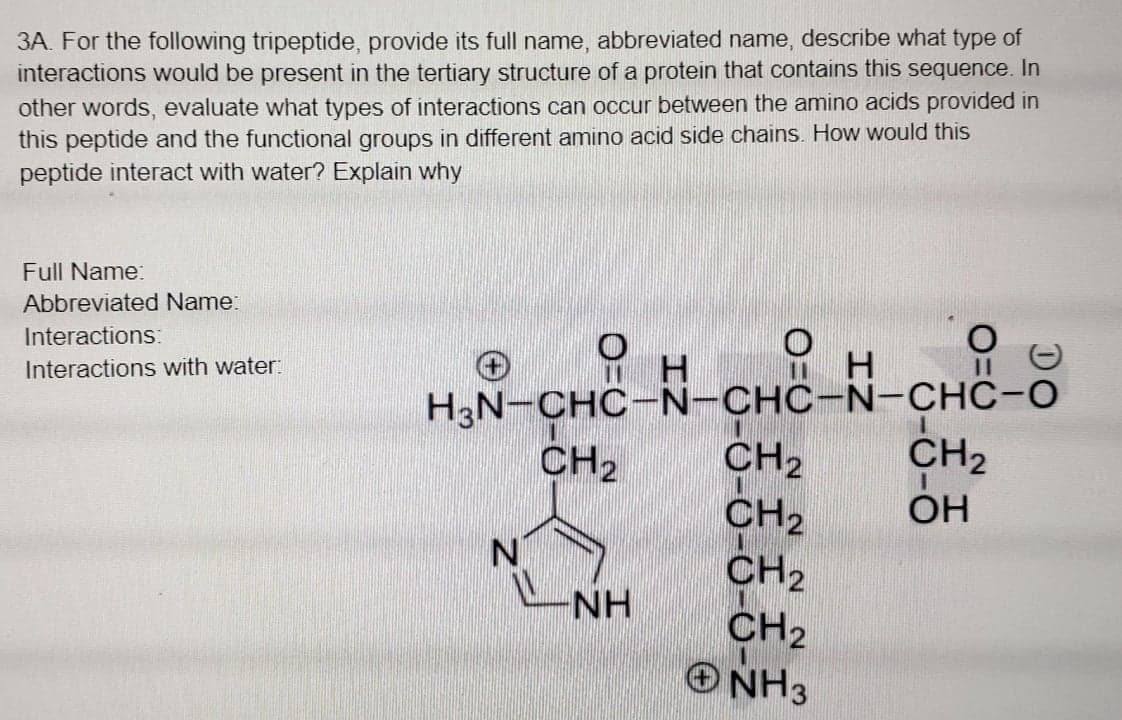
Transcribed Image Text:3A. For the following tripeptide, provide its full name, abbreviated name, describe what type of
interactions would be present in the tertiary structure of a protein that contains this sequence. In
other words, evaluate what types of interactions can occur between the amino acids provided in
this peptide and the functional groups in different amino acid side chains. How would this
peptide interact with water? Explain why
Full Name:
Abbreviated Name:
Interactions:
H.
H.
H3N-CHC-N-CHC-N-CHC-O
CH2
CH2
CH2
CH2
ONH3
Interactions with water:
CH2
CH2
OH
N.
NH
O=0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax