5. cis-[Fe(phen)2(NCS):] is an example of a unique metal complex which undergoes a process known as spin crossover at a specific temperature. The graph below shows a plot of magnetic susceptibility versus temperature. Explain what is happening at approximately 175 K when there is a dramatic change in magnetic susceptibility for the sample. HINT: Start by drawing a d-orbital splitting diagram. (cm/mol K) 2 phen = 100 150 200 250 300 T (K)
5. cis-[Fe(phen)2(NCS):] is an example of a unique metal complex which undergoes a process known as spin crossover at a specific temperature. The graph below shows a plot of magnetic susceptibility versus temperature. Explain what is happening at approximately 175 K when there is a dramatic change in magnetic susceptibility for the sample. HINT: Start by drawing a d-orbital splitting diagram. (cm/mol K) 2 phen = 100 150 200 250 300 T (K)
Chapter26: Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 26.29QAP
Related questions
Concept explainers
Atomic Structure
The basic structure of an atom is defined as the component-level of atomic structure of an atom. Precisely speaking an atom consists of three major subatomic particles which are protons, neutrons, and electrons. Many theories have been stated for explaining the structure of an atom.
Shape of the D Orbital
Shapes of orbitals are an approximate representation of boundaries in space for finding electrons occupied in that respective orbital. D orbitals are known to have a clover leaf shape or dumbbell inside where electrons can be found.
Question
100%
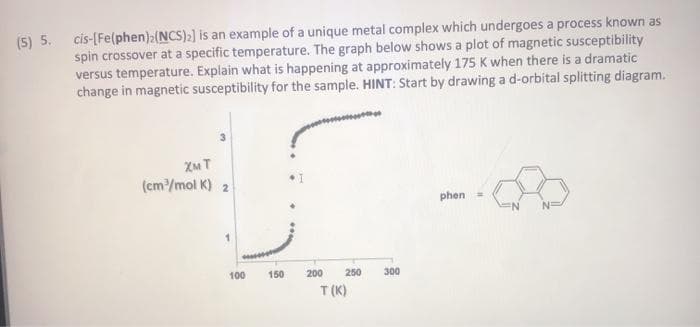
Transcribed Image Text:(5) 5. cis-[Fe(phen):(NCS)) is an example of a unique metal complex which undergoes a process known as
spin crossover at a specific temperature. The graph below shows a plot of magnetic susceptibility
versus temperature. Explain what is happening at approximately 175 K when there is a dramatic
change in magnetic susceptibility for the sample. HINT: Start by drawing a d-orbital splitting diagram.
XM T
(cm/mol K) 2
phen =
100
150
200
250
300
T (K)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning