5th 8th 3rd 7th 6th 7th 2nd Use the data table for the first eight ionization energies of a particular element shown below to predict and explain which element you would expect to show this trend in successive ionization energies. Jonization lonization Energy Number (kJ/mol) 1st 1. Fluorine 2. Oxygen 1,314 3,388 5,300 7,469 10,990 13,327 71,330 84.078 Because 1. core electrons are more strongly attracted to the nucleus than valence electrons. 2. valence electrons release more energy when they are removed from the atom.
5th 8th 3rd 7th 6th 7th 2nd Use the data table for the first eight ionization energies of a particular element shown below to predict and explain which element you would expect to show this trend in successive ionization energies. Jonization lonization Energy Number (kJ/mol) 1st 1. Fluorine 2. Oxygen 1,314 3,388 5,300 7,469 10,990 13,327 71,330 84.078 Because 1. core electrons are more strongly attracted to the nucleus than valence electrons. 2. valence electrons release more energy when they are removed from the atom.
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter7: The Structure Of Atoms And Periodic Trends
Section: Chapter Questions
Problem 69SCQ: Answer the following questions about first ionization energies. (a) Generally ionization energies...
Related questions
Question
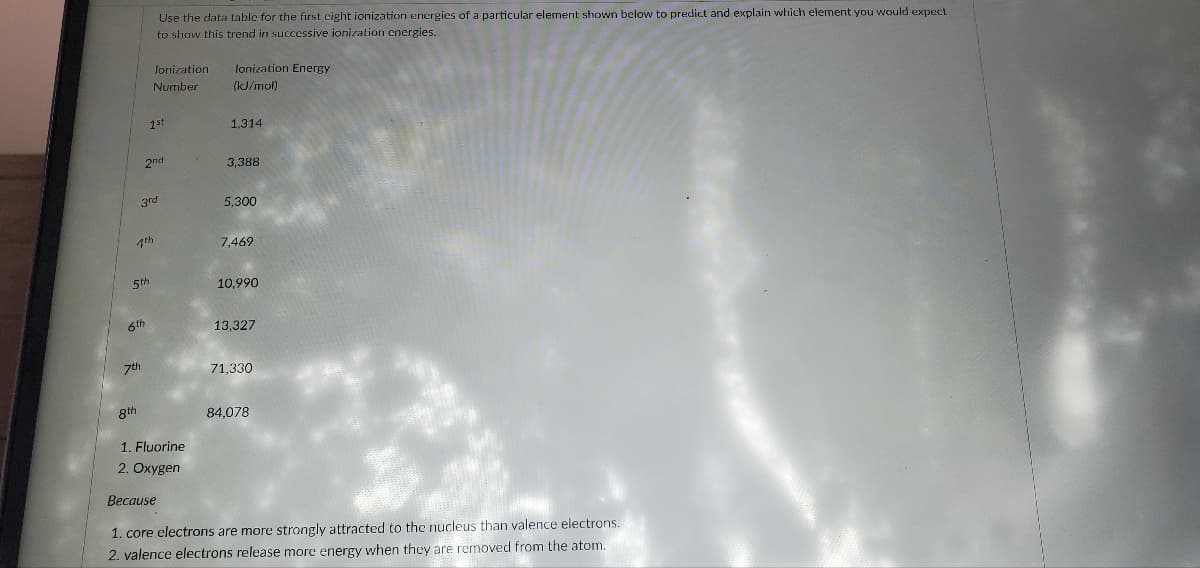
Transcribed Image Text:7th
5th
3rd
6th
7th
8th
2nd
Use the data table for the first eight ionization energies of a particular element shown below to predict and explain which element you would expect
to show this trend in successive ionization energies.
Jonization
Number
1st
1. Fluorine
2. Oxygen
lonization Energy
(kJ/mol)
1,314
3,388
5,300
7,469
10,990
13,327
71,330
84.078
Because
1. core electrons are more strongly attracted to the nucleus than valence electrons.
2. valence electrons release more energy when they are removed from the atom.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning