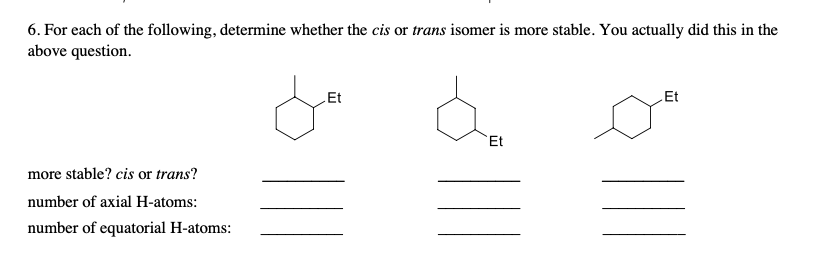
6. For each of the following, determine whether the cis or trans isomer is more stable. You actually did this in the above question. LEt Et Et more stable? cis or trans? number of axial H-atoms: number of equatorial H-atoms:
Q: IX. Rank in order of increasing stability using the less than (<) inequality symbol. Label the…
A: Stability of organic molecules depends on various factors such as molecular interactions,…
Q: least stable chair conformer of the following compound?
A: Required answer is as follows:
Q: 1 ethel 2 isopropylcyclohexane Which one would be a most stable cis?
A: The isomers which are formed by the rotation of single bond is known as conformation isomerism.
Q: Question 5 How many degrees of unsaturation are present in the following molecules: ii C;H,CI i ii…
A:
Q: Convert the following to Newman projections looking down the indicated bond RIGHT to LEFT RIGHT to…
A:
Q: Consider the alkene: (image 1) Is the following a cis-trans isomer, a constitutional isomer, or…
A: constitutional isomers are those in which the molecular formula remains the same in both the case…
Q: Calculate strain energy for the conformer pictured below, using strain energy increments from the…
A:
Q: 3. Explain with example what do you mean by the tesms i somer and conformational isomer ?…
A: We will answer the first one since the exact one was not specified. Please resubmit a new question…
Q: Determine which cyclohexane structure has the MOST energy (is the LEAST stable)?
A:
Q: 4. For the following structures, draw both possible chair conformations, using a ring flip to go…
A: chair conformations of the given trisubstituted cyclohexanes are given below in the attached sheet.
Q: Look at a cyclohexane model: a. Mark the axial bonds. Hold carbon atoms 1, 2, 3, 4, 5, and 6 firmly…
A:
Q: Question attached
A: Introduction: Molecular structure shows the arrangement of atoms in a space. The organic structures…
Q: Draw all constitutional isomers of all-cis ethylmethylisopropylcyclohexane—that is, in which a…
A: Constitutional isomers are compounds that have the same molecular formula and different…
Q: Using Newman projections, draw all of the staggered and eclipsed conformations that result from the…
A:
Q: This problem considers the conformational isomers of 2-methyl-butane shown below. The label for each…
A: In Newman projection, staggered conformation is considered to be the most stable conformation with…
Q: Which other factor along with torsion decide the stability of conformer?
A: Since, you have posted multiple questions, we will solve the first one for you. For the rest to get…
Q: Part B: NH2
A: A question based on molecules that is to be accomplished.
Q: Draw the most stable Newman projection for the following alkanes looking down the C-C bond pointed…
A: The most stable Newman projection for the following alkanes looking down the C-C bond pointed to by…
Q: Interconversion of the staggered and eclipsed conformations of alkanes requires rotation around a -C…
A: The dihedral angle of a molecule provides information about the angle between the groups that reside…
Q: /hich stereoisomer can obtain the least amount of conformational strain, cis- or trans-1,3-…
A: We know that group at axial position in chair form feel more repulsion than equitorial position .
Q: Consider 1,2-dimethylcyclohexane.Q: Which isomer, cis or trans, is more stable and why?
A: The possible isomers of 1,2-dimethylcyclohexane is shown below,
Q: 1,3-dihydroxycyclohexane more stable? , is a cis isomer or trans isomer ?
A:
Q: Answer the following questions and upload your answer. A. Draw the two chair conformations of…
A: A. Trans form is more stable than cis here.
Q: Draw both conformations for 1-ethyl-1-methylcyclohexane and decide which conformation (if any) is…
A:
Q: The equilibrium constant for the conversion of the axial to the equatorial conformation of…
A: a) If the equilibrium constant, K>1 then equilibrium favors towards the product. So, according…
Q: What are the constitutional isomers (illustrated both in condensed and bond lines) for
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: _6. Which term can be used to define a molecule that is cyclic, planar, completely conjugated, and…
A:
Q: 2. For each pair of molecules, circle the term―enantiomers, diastereomers, constitutional isomers,…
A:
Q: Circle the following molecules that have the R configuration
A: R indicates that a clockwise circular arrow that goes from higher priority to lower priority crosses…
Q: 2)- Determine the total number of conformers of the tetra-substituted cyclohexane structure given…
A: Hello. Since your question has multiple sub-parts, we will solve only the first three sub-parts for…
Q: Below are the two chair conformations of 1,1,2-trimethylcyclohexane. What is the approximate…
A:
Q: Given cyclohexane in a chair conformation, construct the more stable conformation of…
A:
Q: Which is more stable: cis- or trans-1-(1-ethylpropyl)-3-methylcyclohexane? Draw the most stable…
A: cis and trans conformer of substituted cyclohexane exist as boat conformer where the two bulky group…
Q: Below you will see a wedge-dash diagram, and an incomplete Newman projection with the designated…
A: To find: complete Newman projection that would give the most stable conformer.
Q: H och
A: In organic chemistry, constitutional isomers or structural isomers are those which have the same…
Q: Draw a model of ethane. Imagine rotating the C-C single bond. Is there a change in the relative…
A: The rotation along the C-C ethane is possible, this rotation can be explained by Newman projections.…
Q: (a) Draw the Newman projection for each molecule shown here, looking down the C-C bond indicated by…
A: Newman projection for the above molecule has to be drawn and the more stable configuration of the…
Q: Based on reaction 1, how many isomers can be formed? Is the isomer cis or trans? The target…
A: The answer is as follows:
Q: ent staggered conformations. Draw them both in Newman projections, tell which is more stable, and…
A:
Q: For 1,2-dichloroethane: ( Q.) Which eclipsed conformation(s) has (have) the lowest energy? Which has…
A: The Newman projections for all eclipsed conformations of 1,2- dichloroethane are shown below along…
Q: Both cis and trans isomers exist for this molecule. Which one is more stable?
A: For this molecule, two structures are possible i.e cis decalin and trans decalin . These are the…
Q: Calculate strain energy for the conformed pictured below, using strain energy increments from the…
A:
Q: In C6H6OH, the bond that undergoes heterolytical change most readily is * С-С С-О С-Н O-H None of…
A:
Q: Organic Chemistry Discuss and compare two possible chair conformations of cis-1,3-dibromocyclohexane…
A:
Q: Sighting along the C2-C3 bond of 2-methylbutane, there are two differ- ent staggered conformations.…
A: Conformations in a molecule refers to the different orientation or positions in which the bonds of…
Q: , draw its and then in the 2. For (1R, 2S, 4R)-4-tert-buyl-1-ethyl-2-methylcyclohexane: draw its…
A: Drawing skeletal structure of (1R,2S,4R)-4-tert-butyl-1-ethyl-2-methylcyclohexane.
Q: I know the answer is correct but I can’t figure out if C is tertiary ? It can’t be secondary because…
A: The reactive intermediate species in which carbon atom forms only three bonds and contains a…
Q: What is the lowest energy 3D conformation of the molecule in the image?
A: A cyclohexane confirmation is three-dimensional shape adopted by a cyclohexane molecule.…
Q: build the secular det erminant bor cyclobutadiene and cofficents and levels
A: Using Huckel molecular orbital to determine secular determinant and energy levels diagram.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- need help. Out of the four isomers shown above (menthol, isomenthol, neomenthol, and neoisomenthol), which isomer do you predict is the most stable? It may help to draw out each isomer in its most stable chair conformer to determine which has the most equatorial groups.Create a conformational analysis using the molecule in the picture. Follow the instruction in the picture when you transform the molecule into its Newman projection. Which conformer is the most unstable? Explain.Consider the Newman projection below. a. Draw a full Lewis structure of this molecule with R1=Me,R2=Et , and R3=iPr . b. Given the sizes of these R groups (R3R2R1) , does the Newman projection above show thelowest potential energy conformation of this bond? If not, draw a Newman projectionshowing the lowest P.E. conformation (sighting down this same bond). c. To draw a Newman projection in the lowest P.E. conformation, the following rule of thumbusually applies: Place the largest group on the front carbon anti to the largest group on theback carbon. Is your answer to the previous question consistent with this rule of thumb?
- Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.Using your model of butane (CH3CH2CH2CH3) , complete the following graph of the anglebetween the two Me groups vs. potential energy. a. Label each Newman projection of butane on the graph with the words staggered, eclipsed, gauche, and anti, as appropriate. (Note that some structures will have more than one label.) b. Draw a wedge and dash bond representation of butane in its lowest P.E. conformation.Create a conformational analysis using the molecule in the picture. Follow the instruction in the picture when you transform the molecule into its Newman projection. Which conformer is the most stable? Explain.
- Which conformation shown below is the most stable conformation of 2-methylbutane?Consider 1,2-dimethylcyclohexane.Q: Which isomer, cis or trans, is more stable and why?translate the bond-line notation structure to the Newman projection by filling int the missing groups (A, B, C, D or E) on the lines in the Newman projection so they match the conformation given in the original structure. Circle if the conformation is a staggered or eclipsed.

