7. Consider the following complexes and enter the requested numbers in (a) and (b): PMes PMes Bu Bu co „CH, но. oc, -CI Me Me oc čo | to čo CH co PMe PMe, Bu" ČN A D E (a) Count electrons around the metal in each complex and enter the numbers in the coesponding lines.
7. Consider the following complexes and enter the requested numbers in (a) and (b): PMes PMes Bu Bu co „CH, но. oc, -CI Me Me oc čo | to čo CH co PMe PMe, Bu" ČN A D E (a) Count electrons around the metal in each complex and enter the numbers in the coesponding lines.
Chapter17: Complexation And Precipitation Reactions And Titrations
Section: Chapter Questions
Problem 17.12QAP
Related questions
Question
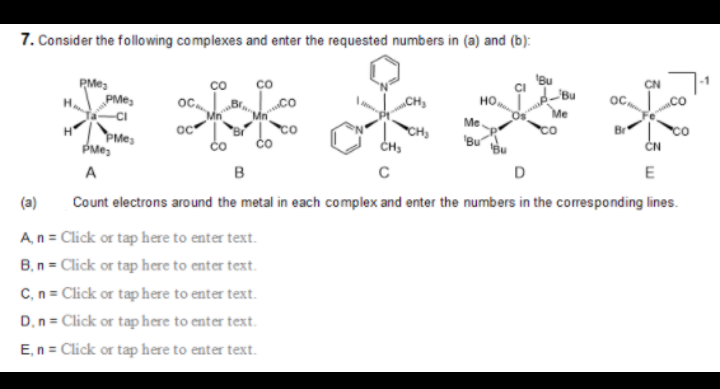
Transcribed Image Text:7. Consider the following complexes and enter the requested numbers in (a) and (b):
Bu
"Bu
PMes
PMe,
-CI
PMes
PMe,
ос,
„CH3
но,
Me
Me
H
oc
"Br
Br
čo
čo
CH,
'Bu
ČN
A
D
E
(a)
Count electrons around the metal in each complex and enter the numbers in the corresponding lines.
An = Click or tap here to enter text.
B. n = Click or tap here to enter text.
C, n = Click or tap here to enter text.
D. n = Click or tap here to enter text.
E,n = Click or tap here to enter text.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



